- *Corresponding Author:
- C. Surushe
Bharati Vidyapeeth’s College of Pharmacy, Navi Mumbai, Maharashtra 400614, 1S.M.B.T College of Pharmacy, Nashik, Maharashtra 422403, India
E-mail: chaitanyasurushe31@gmail.com
| Date of Received | 14 December 2023 |
| Date of Revision | 29 July 2023 |
| Date of Accepted | 14 December 2023 |
| Indian J Pharm Sci 2023;85(6):1586-1593 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
Nanotechnology advancements have resulted in the creation of tailored medicine delivery systems. Because of its potential to address a wide range of unexplored difficulties, nanotechnology-based medication delivery systems are acquiring significant relevance in the modern day. Nanosponges are microscopic sponges the size of a virus (250 nm-1 μm) having cavities that may be filled with a wide range of hydrophobic and hydrophilic medicines also overcome issues such as medication toxicity and low absorption. Nanosponges are solid porous particles with the ability to load medications and other actives into their nanocavity; they can be manufactured in oral, topical, parenteral or inhalation dosage forms. Crosslinking cyclodextrin with carbonyl or di-carboxylate can be used to create nanosponges (Crosslinkers). Another key features of nanosponges includes their aqueous solubility, which allows them to be used efficiently for medications with low solubility. Nanosponges can also be used as a vehicle for enzymes, proteins, vaccines and antibodies.
Keywords
Nanosponges, emulsion solvent diffusion methods, drug loading
Nanotechnology is defined as the manipulation of matter on an atomic, molecular, and supramolecular scale, which includes the design, production, characterization and application of various nanoscale materials in various potential areas, primarily in the field of medicine, to provide novel technological advances. The desired goal of novel drugs (especially nanosponge) to build the advance targeting drug delivery systems was basically achieved. Nanosponge drug delivery systems were originally only available as a topical administration system, but in the 21st century, Nanosponges can now be administered via oral and Intravenous (IV) routes. The nanosponges are a three-dimensional polyester scaffold (backbone) or network that can naturally degrade. To make Nanosponges, these polyesters are combined with a crosslinker in a solution. Nanosponge is a novel type of material made composed of very small particles having a very narrow cavity of a few nanometers. These little spaces can be filled with a variety of materials. These nanosponges have the ability to convey both hydrophilic and lipophilic medication substances, as well as boost the stability of drug substances or compounds that are weakly water soluble. These tiny sponges can circulate throughout the body until they reach a specific target region, where they will attach to the surface and begin to release the medicine in a controlled and predictable manner. The drug will be more effective for a given dosage since it can be released at a specific target spot rather than spreading throughout the body. Nanosponges can be employed as a vehicle for pharmacological properties to improve lipophilic drug water solubility, protect degradable molecules and construct drug delivery systems for routes other than oral administration. The preparation of polymers and crosslinkers is quite straightforward due to their simple chemistry and this technique can readily be scaled up to commercial production levels. Water soluble nanosponges do not break down chemically in water. They are used as a transport fluid when mixed with water. They can be used to cover up disagreeable flavours or to turn liquids into solids. The chemical linkers allow the nanosponges to attach to the target location preferentially. Nanosponges are superior to microsponges in terms of application because their smaller size causes less disruption in the system in which they are used, lowering the likelihood of failure or negative consequences (fig. 1 and fig. 2) [1,2].
Advantages[1-4]
Nanosponge molecules are soluble in water and can be enclosed within the sponge. Drug irritability can be significantly reduced with nanosponges without compromising efficacy. By prolonging the time between doses, nanosponges improve patient compliance. A targeted, site-specific medication delivery method is provided by nanosponge. To avoid the unwanted (bitter) taste nanosponges can be used to conceal medicine tastes that are too bitter and to turn liquid substances into solids. The suitable materials in this technique can act as a protective barrier, protecting the drug from the body's earliest stages of removal. Better stability, enhanced elegance, and greater formulation flexibility are all advantages of nanosponges. Nanosponges are non-toxic, non-allergenic, nonmutagenic, and non-irritating. These are selfsterilizing as their average pore size is 0.25μm, where bacteria cannot penetrate. Extended release continuous action up to 12 h. Compatible with other components and freely flowing (Table 1).
| Polymer | Copolymer | Crosslinker |
|---|---|---|
| Hyper cross-linked polystyrenes, cyclodextrine and its derivatives like methyl ?-cyclodextrine, 2-hydropropyl ?-cyclodextrine | Ethyl cellulose (EC), polyvinyl alcohol (PVA) | Di-phenyl Carbonate (DPC), diarylcarbonate, diisocyanates, pyromelliticanhydride, carbonyl diimidazole, 22-bis (acrylamide) acidic acid and dichloromethane |
Table 1: Materials Used in the Preparation of Nanosponges
Disadvantages
Nanosponges incorporate only tiny molecules. Nanosponges are dependent on loading capacities[1]. Dose dumping may take place in nanosponges[2]. May delay the release.
Methods
Solvent method:
Nanosponges are made using the solvent technique by combining the polymer with polar aprotic solvents like Dimethyl Sulfoxide (DMSO) and Dimethylformamide (DMF). Then, a crosslinker is added to this mixture at 1:4 ratio. The abovementioned reaction needs to be conducted at a temperature of 10° to reflux the solvent's temperature for a duration of 1 h-48 h. The solution is cooled to room temperature when the reaction is finished, and the resultant product is then added to bi-distilled water. The product was recovered by filtering it under vacuum conditions. The substance is next purified by Soxhlet extraction with ethanol and lastly dried to obtain final product[2].
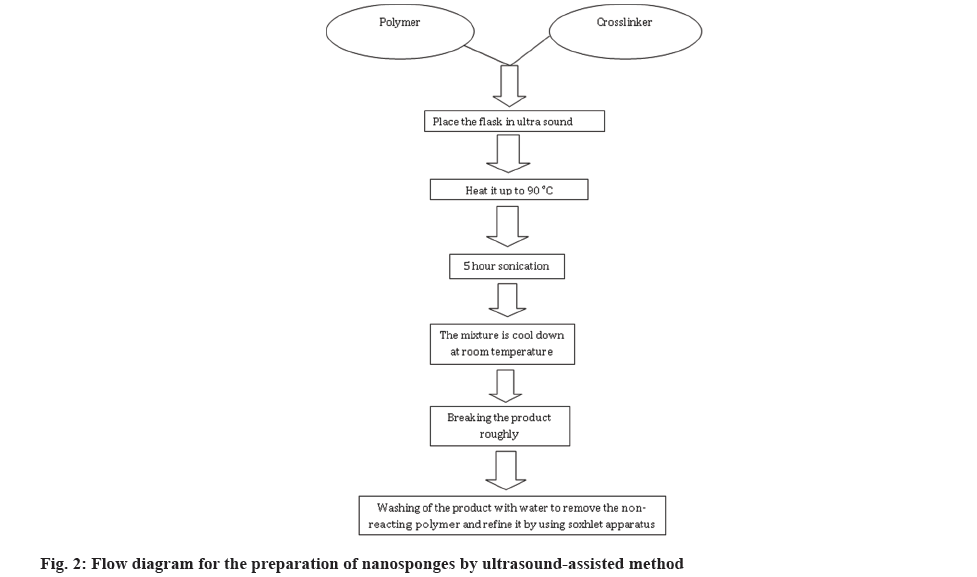
Ultrasound assisted synthesis:
In this method, the crosslinkers and polymers are allowed to react, in the absence of solvents under sonication to produce nanosponge. The combination is sonicated for 5 h while the flask is submerged in an ultrasonic bath that is filled with water and heated to 90°. The product is next broken into rough bits when the mixture has cooled to room temperature. In order to produce nanosponges, the non-reacting polymer is finally eliminated by washing the product with water and refining it using a Soxhlet apparatus (ethanol)[5].
Emulsion solvent diffusion method:
This approach creates nanosponges by mixing ethyl cellulose and polyvinyl alcohol in various ratios or amounts. This approach employs both dispersed and continuous phases. Ethyl cellulose and the drug collectively forms the dispersed phase, which is then combined with 20 ml of dichloromethane and some polyvinyl alcohol (PVA) to create the continuous phase (aqueous). The mixture is then agitated for around 2 h at a speed of 1000 rpm. The finished product, or nanosponges, is obtained by filtration. The product is dried in the oven at a final temperature of 400°[2].
Nanosponges made from hyper cross-linked β-cyclodextrins:
Nanosponges are made from materials that make non-porous molecules that are carriers called cyclodextrins for drug release. These cyclodextrins are hyper-cross-linking substances that create several nanoscale networks or can even take the form of a sphere with countless networks of protein channels, pores, etc. Based on the chemicals they contain, these cross linkers stabilise the sponge and give it a certain surface charge density, porosity, and pore size. Cross linkers aid in maintaining Nano sponges at various acidic and even neutral pH levels[6].
Drug loading:
Nanosponges need to be pre-treated in order to produce particles smaller than 500 nm. The nanosponges are dissolved or suspended in water to attain this range. To stop the buildup, the nanosponges in suspension are aggressively sonicated. A colloidal fraction is created by centrifuging the suspension. Using a freeze dryer, the sample is dried after the supernatant has been removed. Nanosponges are synthesized as an aqueous solution. For the complexation to occur, an excess amount of medication is added to the suspension and continuously swirled for a set period of time. After complexation has occurred, the uncomplexed drug is removed from the complexed drug by centrifugation. The freeze dryer or solvent evaporation are two methods for producing the solid crystals of the nanosponges. In the complexation of the medication, this Solid Crystal structure plays a key role. When compared
Mechanism
The active ingredient is given to the vehicle in an encapsulated form since nanosponges have an open structure and lack a continuous membrane around them. From the particles into the vehicle, the encapsulated active ingredient can travel freely until the vehicle becomes saturated and equilibrium is reached. The vehicle carrying the active ingredient becomes unsaturated as soon as the product is applied to the skin, disrupting the balance. Thus, until the vehicle is either absorbed or dried, active compounds from nanosponge particles start to flow into it. The release of active substance to skin continues for a considerable amount of time even after the retention of the nanosponge particles on the stratum corneum of the skin[2]. The nanosponges are encapsulated in various routes of drug administration such as topical, inhalational, parenteral or oral dosage forms. In oral route, they are consumed as tablets or capsules in which there may be a matrix of lubricants, excipients, diluents and anticaking agents. In parenteral route, the drugs may be composed of aqueous solutions, saline, and sterile water[7].
Applications
Long-term delivery system:
One of the most often utilised antiviral medications for the treatment of herpes simplex virus infection is acyclovir. Its digestion by the gastrointestinal tract occurs slowly, insufficiently, and with great variability. The acyclovir in vitro release profile from several nano sponge types demonstrated prolonged drug release. After 3 h of treatment, carb-nanosponges and nanosponges released acyclovir in amounts of around 22 % and 70 %, respectively. Since there was no first burst effect seen, the medication was not adsorbed on the surface of the nanosponges[8,9].
Solubility enhancement:
Itraconazole is a Biopharmaceutics Classification System (BCS) class II medication with low bioavailability and a dissolving rate restriction. Thus, the use of nanosponges increased the drug's solubility by a factor of over 27. When copolyvidonum was introduced as a supporting component, the solubility was shown to be 55 times greater. Nanosponges increase the medication's solubility by either disguising the hydrophobic groups of itraconazole, enhancing the drug's wetting ability, or reducing the crystallinity of the drug[10].
Drug delivery:
Nanosponges are excellent for carrying medications that are not soluble in water due to their nanoporous nature (BCS class-II drugs). These complexes can be used to hide disagreeable odours, turn liquid substances into solids, and enhance the pace, solubility, and stability of medication dissolution. Compared to direct injection, cyclodextrin-based nanosponges are said to transport the medication to the target spot three to five times more efficiently[11]. By loading into the nanosponges, medications that are particularly important for formulation in terms of their solubility may be properly supplied.
The nanosponges can be made into dosage forms for oral, parenteral, topical, or inhalation use. They are solid by nature. The complexes may be disseminated in a matrix of excipients, diluents, lubricants and anticaking agents appropriate for the manufacture of capsules or tablets for oral delivery[12]. The compound can easily be transported in sterile water, saline, or other aqueous solutions for parenteral administration[12]. They can be successfully integrated into topical hydrogel for topical delivery[6,13].
A class II medication having a dissolution raterestricted bioavailability is Telmisartan (TEL). The formulation of the nanosponge included TEL. The beta-CD complex of TEL was compared to plain TEL and the nanosponge complexes of TEL in terms of vitro dissolution and saturation solubility. Inclusion complexes made from nanosponge and NaHCO3 (Sodium bicarbonate) showed the greatest solubility and in vitro drug release. The anticancer medication paclitaxel has a low solubility in water. Beta-CD based nanosponges are a substitute for the traditional formulation in cremophor since cremophor reduces the tissue penetration of paclitaxel. The nanosponge formulation significantly improves the biological impact of paclitaxel in vitro. An antifungal treatment for dermatophytosis and skin infections is econazole nitrate. Skin application of econazole has little adsorption. Thus, the solvent diffusion approach is used to create econazole nitrate nanosponges, which are then loaded as hydrogels[14].
Enzyme immobilization:
The enzyme has frequently been stabilised using nanosponges. When compared to cyclodextrin, Cyclodextrin Nanosponge (CD-NS) exhibits substantially higher inclusion constants and is an appropriate support for enzyme immobilisation. They aid in maintaining the stability and catalytic efficiency of the immobilised enzymes. Enzyme immobilisation is crucial for enzyme recycling and makes it easier to separate and recover the generated products. It also increases the biocatalysts' thermal and operational stability. The high catalytic activity of various lipases from Pseudomonas flurescens adsorbed on cyclodextrin-based nanosponge was also examined by Boscolo et al.[15]. Numerous industrial applications include the hydrolysis of triacylglucerols and trans esterification processes, both of which are catalysed by lipases.
Protein delivery:
The preservation of the original protein structure during the formulation process and during longterm storage represents a significant hurdle in the creation of protein formulations. Swaminathan et al.[16] explored novel swellable poly nanosponges based on cyclodextrin. They discovered very strong swelling capacity that remained steady for 72 h through research on water intake. As a model protein, bovine serum albumin was employed and it was integrated into the finished nanosponge. Protein stability was raised and its enhanced swelling property was seen. The lactone ring opens up and transforms into an inactive carboxylate form at physiological pH. Camptothecin is fused in nanosponges, resulting in a longer release profile in an active state that prevents the hydrolysis of the lactone form and leads to increased stability[16].
Protective agent from light or degradation:
It is possible to encapsulate gamma-oryzanol in the form of a nanosponge, which exhibits good photo-degradation protection. Gamma oryzanol is a naturally occurring antioxidant and ferulic acid combination that is primarily utilised to stabilise food and pharmaceutical raw materials. Due to its high instability and photogegradation, its applicability is restricted[17].
Carrier for biocatalyst:
The biological and therapeutic fields can also make use of proteins, peptides, enzymes and their derivatives. While DNA and oligonucleotides are utilised in gene therapy, proteolytic enzymes can be employed to treat cancer or type I mucopolysaccharidosis. These molecules' administration comes with a number of drawbacks and issues. Because of their large molecular size, hydrophilic nature, degree of ionisation, high surface charge, chemical and enzymatic instability and limited permeability across mucous membranes, the majority of protein medicines are poorly absorbed through biological membranes. Protein molecules may be quickly removed from the blood after IV delivery, bind to plasma proteins and be vulnerable to proteolytic enzymes. The issue with oral administration is bioavailability. There are other methods for therapeutic usage, such raising the dosage or utilising absorption boosters, which may result in toxicity issues[18]. There are several mechanisms for transporting enzymes and proteins, including hydrogels, nano and microparticles, and liposomes. Carriage in a certain system can alter the pharmacokinetics of proteins, prevent them from degrading, and increase their in vivo stability. Cyclodextrinbased nanosponges have now been discovered to be a very effective carrier for the adsorption of proteins, enzymes, antibodies and macromolecules. It is possible to maintain enzyme activity and efficiency, prolong operation, increase the pH and temperature range of activity, and perform continuous flow operations, in particular when enzymes are utilised. Additionally, by adsorbing or encapsulating proteins and other macromolecules in CD-NS, they can be transported[18].
Gas delivery system:
Hypoxia, the lack of a sufficient oxygen supply, is linked to a number of diseases, including cancer and inflammatory diseases. A nanosponge formulation for topical oxygen delivery was created by Cavalli et al.[18]. In vero cells, the safety of nanosponge was investigated. Using a CD-NS hydrogel combination technology, oxygen penetration across a silicone membrane was investigated. For the encapsulation of 1-methylcyclopropene, oxygen, and carbon dioxide, Scientist described the preparation of CDNS utilising carbonildiimidazole cross-linker[19].
Cancer therapy:
The distribution of anticancer drugs is now one of the most difficult tasks in the pharmaceutical industry because of their poor solubility. According to one report, direct injection is three times less efficient than nanosponge’s complex at slowing tumour development. The complex of the nanosponge loads a medication and exposes a targeting peptide that adheres firmly to the tumour receptor's top layer of radiation-induced cells. When nanosponges come into contact with a tumour cell, they adhere to its surface and begin to release medication molecules. Targeting medication delivery has the benefit of achieving a more potent therapeutic impact at a lower dose and with fewer adverse effects[20].
Oral delivery:
For oral delivery as capsules or tablets, the complex can be disseminated in a matrix comprising diluents, excipients, lubricants and anti-caking agents. Nanosponges can increase the wetting and solubility of molecules that have low water solubility. The medications may be molecularly disseminated inside the nanosponge structure and subsequently released as molecules, eliminating the need for disintegration. As a result, the drug's perceived solubility can be boosted. Many formulation and bioavailability issues may be overcome by increasing a substance's solubility and dissolving rate, and nanosponges can significantly increase medication solubility. BCS class II medicines have relatively poor solubility and are thus great candidates for nanosponges[21]. To produce nanosponges for use in an oral medication administration system, acetyl salicylic acid, a Nonsteroidal Anti-Inflammatory Drug (NSAID) classed as a BCS class II agent, was utilised. When taken orally, it creates a nanosponge system with holes that speed up the solubilization of medications with low water solubility by trapping them in the pores[22].
Topical delivery:
Topically, nanosponge components can be administered in the form of a gel or cream. Resveratrol-loaded nanosponges were thought to improve drug penetration on pig skin in vitro. The capacity of nanosponges to boost solubility at the skin's surface may also be linked to their ability to improve guest molecule absorption by the skin[23].
Chemical sensors:
Titanium nanosponges, a particular kind of "metal oxides," are utilised in the very sensitive detection of hydrogen as chemical sensors. Since there is initially no point of contact, there is less resistance to electron transport, which leads to a greater level of 3D interconnect and more sensitive nanosponge materials to H2 gas[24].
Antiviral application:
Nanosponges are administered via the nasal and pulmonary routes. It offers selectivity to target viruses that may cause respiratory tract infection, such as influenzavirus and rhinovirus and deliver antiviral drugs on Ribonucleic Acid (RNA) to the lungs or nasal pathway using nanocarriers. Zidovudine and Saquinavir are two medications utilised as nanocarriers[25].
Oxygen delivery system:
Oxygen delivery system characterised using cyclodextrins of the molecular weights alpha, beta, and gamma that are suspended in water and become saturated with water. With the aid of a nanosponge/ hydrogel combination, a silicone membrane may likewise be employed to let oxygen through[26].
SARS-CoV-2 Management:
Nanosponges made from human macrophages or lung type II epithelial cells are a sufficient attractant for the SARS-CoV-2 virus and may be eliminated after capture. As a result, it was employed to create SARS-CoV-2 preventive strategies[27]. The researchers created two types of cellular nanosponges based on the existing structure of SARS-CoV-2; human lung epithelial type II cell Nanosponge (epithelial-NS) and human Macrophage Nanosponge (M-NS). Nanosponges carried the same receptors on which viruses rely for entrance and it was hypothesised that after interacting with these nanosponges, the viruses would be unable to enter the cell[7].
Factors Affeting Formulation of Nanosponge
Degree of substitution:
The complexation ability of the nanosponge may be strongly influenced by the kind, quantity, and location of the substituent on the parent molecule[28].
Method of preparation:
The complexation may be impacted by the drug's loading into the nanosponge formulation. The complexation may be impacted by the type of the medication and polymer. Freeze drying has proven to be a more productive approach for pharmacological complexation in many instances[29].
Temperature:
Drug complexation may be impacted by temperature changes. Due to a potential reduction in drug nanosponge contact forces, van der waals forces, and hydrophobic forces with rising temperature, the apparent stability of the nanosponge complex diminishes with temperature[29].
Type of drug:
The following qualities for drug compounds that will be complexed with nanosponges should be present[22]. Water solubility should be less than 10 mg/ml. No more than five condensed rings should be present in the medication molecule structure. Less than 250° should be the melting point. 100 to 400 Da is the required molecular weight range.
Type of polymer and crosslinkers:
The choice of an appropriate polymer affects both the production and performance of nanosponge. The nanosponge's cavity or pore size should be able to fit a medication molecule of the appropriate size[22]. Crosslinkers aid in the formation of a 3D structure of nanosponges. The amount of crosslinker utilized affects drug entrapment as well as organ targeting[30]. The crosslinker utilised determines whether the nanosponge is soluble in water or any other solvent. Epichlorohydrin will be used as a crosslinker to create hydrophilic nanosponges. The benefit of utilising hydrophilic nanosponges in drug delivery is that it enhances drug absorption across biological membranes and is a valuable transporter for pharmaceuticals in order to produce quick release formulations[31-33].
Conclusion
Novel drug delivery systems are being researched extensively, with nanosponges being one of the most successful, as they may carry either lipophilic or hydrophilic drugs and release them at the target location in a controlled and predictable manner. The particle size and release rate may be controlled by adjusting the polymer-to-crosslinker ratio. Nanosponges facilitate the release of insoluble medicines while protecting the active moieties from physicochemical degradation. They are built on Nano, polymer-based spheres that may suspend or entrap a wide range of chemicals before being mixed into a prepared product such as a gel, lotions, cream, ointments, liquid, or powder. This method allows for the trapping of substances, resulting in fewer adverse effects, higher stability, increased elegance, and increased formulation flexibility. Nanosponges can be effectively incorporated into topical drug delivery systems for dosage form retention on skin, as well as used for oral drug delivery using bio erodible polymers, particularly for colon specific delivery and controlled release drug delivery systems, thus improving patient compliance by providing site specific drug delivery systems and prolonging dosage intervals.
Conflict of interest:
The authors declare that this article has no conflict of interest.
References
- Kaivalya IR, Prasad D, Sudhakar M, Bhanja SB, Tejaswi M. A review on nanosponges. Int J Recent Sci Res 2020;11(1):36878-84.
- Bhowmik H, Venkatesh DN, Kuila A, Kumar KH. Nanosponges: A review. Int J Appl Pharm 2018;10(4):1-5.
- Uday B, Manvi FV, Kotha R. Recent advances in nanosponges as drug delivery system. Int J Pharm Sci Nanotechnol 2013;6(1):1935-44.
- Vishwakarma P, Choudhary R. Microsponges: A novel strategy to control the delivery rate of active agents with reduced skin irritancy. J Drug Deliv Ther 2019;9(6-s):238-47.
[Crossref] [Google Scholar] [PubMed]
- Arshad K, Khan A, Bhargav E, Reddy K, Sowmya C. Nanosponges: A new approach for drug targeting. Int J Adv Pharm Res 2016;7(3):381-96.
- Sharma R, Walker RB, Pathak K. Evaluation of the kinetics and mechanism of drug release from econazole nitrate nanosponge loaded carbapol hydrogel. Indian J Pharm Edu Res 2011;45(1):25-31.
- Girigoswami A, Girigoswami K. Versatile applications of nanosponges in biomedical field: A glimpse on SARS-CoV-2 management. Bionanoscience 2022;12(3):1018-31.
[Crossref] [Google Scholar] [PubMed]
- Ansari KA, Vavia PR, Trotta F, Cavalli R. Cyclodextrin-based nanosponges for delivery of resveratrol: In vitro characterisation, stability, cytotoxicity and permeation study. AAPS Pharmscitech 2011;12:279-86.
[Crossref] [Google Scholar] [PubMed]
- Lembo D, Swaminathan S, Donalisio M, Civra A, Pastero L, Aquilano D, et al. Encapsulation of Acyclovir in new carboxylated cyclodextrin-based nanosponges improves the agent's antiviral efficacy. Int J Pharm 2013;443(1-2):262-72.
[Crossref] [Google Scholar] [PubMed]
- Mognetti B, Barberis A, Marino S, Berta G, De Francia S, Trotta F, Cavalli R. In vitro enhancement of anticancer activity of paclitaxel by a Cremophor free cyclodextrin-based nanosponge formulation. J Incl Phenom Macrocycl Chem 2012;74:201-10.
- David F. Salisbury. Nanosponge drug delivery system more effective than direct injection. 2010.
- Alongi J, Poskovic M, Frache A, Trotta F. Role of ?-cyclodextrin nanosponges in polypropylene photooxidation. Carbohydr Polym 2011;86(1):127-35.
- Sharma R, Pathak K. Polymeric nanosponges as an alternative carrier for improved retention of econazole nitrate onto the skin through topical hydrogel formulation. Pharm Dev Technol 2011;16(4):367-76.
[Crossref] [Google Scholar] [PubMed]
- Tejashri G, Amrita B, Darshana J. Cyclodextrin based nanosponges for pharmaceutical use: A review. Acta Pharm 2013;63(3):335-58.
[Crossref] [Google Scholar] [PubMed]
- Boscolo B, Trotta F, Ghibaudi E. High catalytic performances of Pseudomonas fluorescens lipase adsorbed on a new type of cyclodextrin-based nanosponges. J Mol Catal B Enzym 2010;62(2):155-61.
- Swaminathan S, Cavalli R, Trotta F, Ferruti P, Ranucci E, Gerges I, et al. In vitro release modulation and conformational stabilization of a model protein using swellable polyamidoamine nanosponges of ?-cyclodextrin. J Incl Phenom Macrocycl Chem 2010;68:183-91.
- Minelli R, Cavalli R, Ellis L, Pettazzoni P, Trotta F, Ciamporcero E, et al. Nanosponge-encapsulated camptothecin exerts anti-tumor activity in human prostate cancer cells. Eur J Pharm Sci 2012;47(4):686-94.
[Crossref] [Google Scholar] [PubMed]
- Selvamuthukumar S, Anandam S, Krishnamoorthy K, Rajappan M. Nanosponges: A novel class of drug delivery system-review. J Pharm Pharm Sci 2012;15(1):103-11.
[Crossref] [Google Scholar] [PubMed]
- Lee CL, Wu CC, Chiou HP, Syu CM, Huang CH, Yang CC. Mesoporous platinum nanosponges as electrocatalysts for the oxygen reduction reaction in an acidic electrolyte. Int J Hydrog Energy 2011;36(11):6433-40.
- Bhowmik H, Venkatesh DN, Kuila A, Kumar KH. Nanosponges: A review. Int J Appl Pharm 2018;10(4):1-5.
- Ghurghure SM, Pathan MS, Surwase PR. Nanosponges: A novel approach for targeted drug delivery system. Int J Chem Studies 2018;2(2):78-92.
- Vyas A, Saraf S, Saraf S. Cyclodextrin based novel drug delivery systems. Journal of inclusion phenomena and macrocyclic chemistry. Nanomater Clin Appl 2008;62:23-42.
- Shah AA, Kehinde EO, Patel J. An emerging era for targeted drug delivery: Nanosponges. J Pharm Res Int 2021;33(32A):153-60.
- Kfoury M, Landy D, Fourmentin S. Characterization of cyclodextrin/volatile inclusion complexes: a review. Molecules 2018;23(5):1204.
[Crossref] [Google Scholar] [PubMed]
- D'Emanuele A, Dinarvand R. Preparation, characterisation, and drug release from thermoresponsive microspheres. Int J Pharm 1995;118(2):237-42.
- Alfred M, James D, Camarata A. Physical chemical principles in the pharmaceutical sciences. Physical Pharmacy Lea and Febiger Washington Square. 1983;600.
- Tharayil A, Rajakumari R, Kumar A, Choudhary MD, Palit P, Thomas S. New insights into application of nanoparticles in the diagnosis and screening of novel coronavirus (SARS-CoV-2). Emergent Mater 2021;4:101-17.
[Crossref] [Google Scholar] [PubMed]
- Challa R, Ahuja A, Ali J, Khar RK. Cyclodextrins in drug delivery: An updated review. AAPS Pharmscitech 2005;6:E329-57.
[Crossref] [Google Scholar] [PubMed]
- Sinha VR, Anitha R, Ghosh S, Nanda A, Kumria R. Complexation of celecoxib with ??cyclodextrin: Characterization of the interaction in solution and in solid state. J Pharm Sci 2005;94(3):676-87.
[Crossref] [Google Scholar] [PubMed]
- Sherje AP, Dravyakar BR, Kadam D, Jadhav M. Cyclodextrin-based nanosponges: A critical review. Carbohydr Polym 2017;173:37-49.
[Crossref] [Google Scholar] [PubMed]
- Tiwari K, Bhattacharya S. The ascension of nanosponges as a drug delivery carrier: Preparation, characterization, and applications. J Mater Sci Mater Med 2022;33(3):28.
[Crossref] [Google Scholar] [PubMed]
- Shivani S, Poladi KK. Nanosponges-novel emerging drug delivery system: A review. Int J Pharm Sci Res 2015;6(2):529.
[Crossref] [Google Scholar] [PubMed]
- Shringirishi M, Prajapati SK, Mahor A, Alok S, Yadav P, Verma A. Nanosponges: A potential nanocarrier for novel drug delivery-a review. Asian Pacific J Tropic Dis 2014;4:S519-26.