- *Corresponding Author:
- K. Nagarajan
Alkem Laboratories Ltd., R and D Centre, Industrial Estate 4th Phase, Bangalore-560 058,India
E-mail: dknb69@yahoo.com
| Date of Submission | 16 October 2013 |
| Date of Revision | 24 December 2013 |
| Date of Acceptance | 30 December 2013 |
| Indian J Pharm Sci, 2014;76(1):92-95 |
Abstract
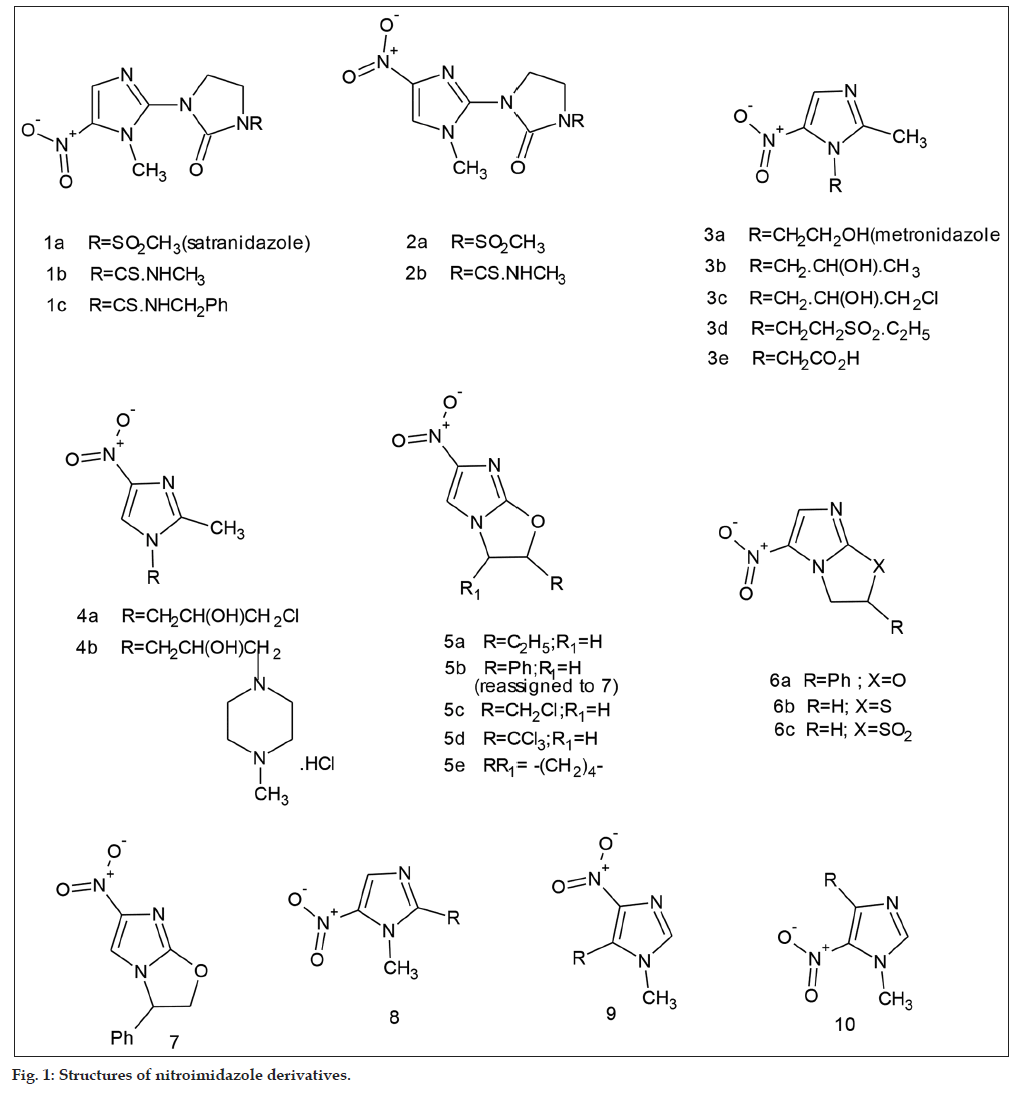
A number of mono and bicyclic nitroimidazoles were screened for in vitro antileishmanial activity. Among these, compounds belonging to the class of nitroimidazo[2,1-b]oxazoles showed moderate to good activity. This class of compounds had been reported previously to have pronounced antitubercular activity, particularly CGI17341 (5a). In the present study (5a) and (5d) and (7) were found to be more potent antileishmanials in vitro than the standard and less toxic in relation to a reference compound. (7) Was earlier formulated to have the phenyl group located on C-2(5b).
Keywords
Nitroimidazoles, antileishmanial activity, nitroimidazooxazoles, satranidazole
Nitroimidazoles, known to have a wide antimicrobial spectrum, are particularly potent against amoeba, giardia and trichomonas [1,2]. Our earlier extensive foray into the medicinal chemistry of nitroimidazoles resulted in the development of satranidazole (satrogyl®), a potent antiamoebic and antitrichomonal drug with clear superiority over the standard drug metronidazole (3a) [3]. (1a) and its congeners had also potent antianaerobic activity superior to that of (3a) [4]. A further outcome of our research was the discovery of significant antitubercular activity in a series of 2,3-dihydro-6-nitroimidazo [2,1-b]oxazoles among which CGI17341 (5a) was the most potent [5,6] that inspired the development of two molecules, which are in clinical trials now, nitroimidazooxazole OPC67683, delamanid [7,8] and nitroimidazooxazine, PA824 [9,10].
Leishmaniasis is a worldwide disease caused by protozoan parasites of the genus of leishmania, which cause a range of diseases in humans ranging from disfiguring cutaneous lesions (CL) to visceral leishmaniasis (VL) [11]. Despite the widespread occurrence and severity of the disease, suitable drugs with a good toxicity profile are not available [12]. In a compounds mining effort, the availability of a library of nitroimidazoles and their ring condensed versions with us attracted the attention of Drugs for Neglected Diseases (DNDi) who undertook to explore their potential for activity against leishmaniasis. Accordingly, about 70 compounds were screened for this activity.
The collection included mainly those that had been investigated for antiamoebic activity earlier and described in our structure–activity relationship paper [3], having the general structures (1-4) and (8-10) (fig. 1). Specially to be mentioned are satranidazole (1a), its 4-nitro analogue (2a), standard antiamoebic nitroimidazoles, secnidazole (3b), ornidazole (3c) and tinidazole (3d). Another group of particular interest for this communication are 2,3-dihydro- 6-nitroimidazo [2,1-b ]oxazoles (5) to which the antitubercular compound (5a) belongs.
The isomeric 2,3-dihydro-3-phenyl-5-nitroimidazo [2,1-b ]oxazole (6a) and two examples of the corresponding imidazothiazoles (6b) and (6c) formed part of this study. Also tested were five derivatives of 2-amino-6-nitrobenzothiazole. The collection had further the following compounds: four mono and dinitropyrazoles and 2,5-dinitrophenyl piperazines having CH3, CO2C2H5 or CON(C2H5)2 group on N(4).
Nitroimidazoles (5a) and (7) previously formulated as (5b) were also resythesised by the earlier procedure [5].
Compound (5a), melting point (m.p.) 160–161°, 1H NMR (400 MHz, CDCl3): δ 7.53 (DMSO-d6, 8.15) (s, 1H), 5.27 (q, J=7 Hz, 1H), 4.37 (dd, J=10.2, 8.3 Hz, 1H), 3.93 (dd, J=10.2,7.4, 1H), 1.9–2.1 (m, 2H), 1.08 (t, J=7.4, 3H); 13C NMR (100.6 MHz, DMSO-d6); δ 156.6, 146.2, 116.5, 89.6, 48.4, 27.2, 9.1; m+ 184.16
Compound (7), m.p. 198-200°, 1H NMR (400 MHz, CDCl3): δ 7.45–7.5 (m, 3H), 7.42 (DMSO-d6, 8.20) (s, 1H), 7.2-7.3 (m, 1H), 5.58 (t, J=8 Hz, 1H), 5.36 (t, J=8.6 Hz, 1H), 4.84 (t, J=8.6, 8 Hz, 1H); 13CNMR (100.6 MHz, DMSO-d6); δ 157.1, 146.2, 127.0, 129.6, 129.7,116.5, 82.2, 59.4; m+ 232.2.
The resynthesis was undertaken for detailed biological studies and also for structure confirmation. This was required because their interesting antitubercular and antileishmanial activity made them important leads for developing new drugs and their route of synthesis indicated uncertainty in the location of the nitro and alkyl/aryl substituents [5]. We carried out extensive 1H, 13C and HMBC spectral studies. While the location of the NO2 group at position 6 in both molecules seemed to be secure, that of the C2H5 group in (5a) and of the phenyl group in the analogue could not be established due to lack of correlation information in the Heteronuclear multiplebond correlation (HMBC) spectra. Single crystal X-ray studies (T.N. Guru Row and Sajesh Thomas. Private Communication) confirmed the structure of (5a) (C2H5group on C-2) while they revealed that the phenyl group had to be located on C-3 as in (7) and not on C-2 as in (5b) as postulated earlier [5].
Activity against Leishmania donovani (axenic) was determined according to the method of Cunningham [13] and L. donovani (macrophage) according to the method of Yang et al . [14]. Miltefosine was the reference drug. Cytotoxicity was assessed by the procedure described by Page et al. [15], the comparative drug being podophyllotoxin. Compounds showing less than 50% inhibition of the axenic culture at the screening dose of 0.8 μg/ml were considered negligibly active and hence of no interest. From the medicinal chemistry point of view, we note that satranidazole (1a), its 4-nitroisomer (2a), other standard antiamoebic drugs, secnidazole (3b), ornidazole (3c), tinidazole (3d) and the 4-nitroisomer, (4a) fall into this category. The IC50 of eight compounds passing the screening test and cytotoxicity are recorded (Table 1) and in vivo data activity against L. donovani for 5a is provided in Table 2.
| Compound | L. donovani (axenic) IC50 (µg/ml) | L. donovani (macrophage) IC50 (µg/ml) | Cytotoxicity L6IC50(µg/ml)>90 |
|---|---|---|---|
| 2b | 0.318 | >90 | |
| 5a | 0.048 | 0.041 | >90 |
| 5c | 0.594 | 88.3 | |
| 5d | 0.045 | 0.22 | 73.3 |
| 5e | 0.591 | >90 | |
| 6c | 0.406 | 1.405 | |
| 7 | 0.081 | 0.42 | >90 |
| 8(R=1‑benzimidazolyl) | 0.476 | >90 | |
| 8(R=2‑methyl‑1, 3, | 0.235 | 1.19 | |
| 4‑thiadiazolyl‑2‑amino) | |||
| Miltefosine | 0.169 | 0.8 | - |
| Podophyllotoxin | - | - | 0.009 |
The IC50 of eight compounds passing the screening test and cytotoxicity
Table 1: In vitro activity against L. Donovani And cytotoxicity
| Compound | Dose | %Inhibition | Group weight% change |
|---|---|---|---|
| 5a | 50 mg/kg i.p.×5 | 43.9 | 1.6 |
| SbV (pentostan) | 15 mg/kg s.c.×5 | 47.2 | 9.2 |
SbV=Pentavalent antimonials, i.p=intraperitoneal injection, s.c=subcutaneous injection
Table 2: In vivo activity against L. Donovani
Compounds 2b (analogue of satranidazole), (6c) [2,3-dihydroimidazo(2,1-b)thiazole dioxide], (8)(R=1- benzimidazolyl) and (8)(R=2-methyl-1,3,4-thiadiazolyl- 2-amino) had modest IC50 values in the ‘axenic’ test but were less potent than miltefosine. Five compounds having the nitroimidazooxazole scaffold had appreciable activity but among these, (5c) and (5e) were less active than the standard. Compound (5a) with an ethyl group and (5d) with a CCl3 group were about 3.5 times more potent than standard while (7) with a phenyl group at C-3 was twice as active. In the macrophage assay, (5a) had 20 times the potency of the standard, (5d) about 3.5 times and (7) about 2 times. (5a) and (7) were not cytotoxic below 90 μg/ml compared with a figure of 0.009 for podophyllotoxin while the IC50 of (5d) in this test was 73.3. These three nitroimidaoxazoles have been taken up for in vivo and genotoxicity studies. The results will be published elsewhere.
The present study reveals that 2,3-dihydro-6- nitroimidazo [2,1-b]oxazoles like the antitubercular CGI 17341( 5a) represent important novel leads for antileishmanial activity.
Acknowledgements
The authors are grateful to Marcel Kaiser and Rito Brun, Swiss TPH, Basel for their help in biological screening and to Sandesh Jatindranath and B. Ugarkar for 1H and 13C NMR data.
References
- Nair MD, Nagarajn K. Nitroimidazoles as chemotherapeutic agents. In: Jucker E, editors. Progress in Drug Research. Vol. 27. Basel: Birkhauser; 1983. p. 163-252.
- Mital A. Synthetic nitroimidazoles: Biological activities and mutagenicity relationships. Sci Pharm 2009;77:497-520.
- Nagarajan K, Arya VP, George T, Nair MD, Sudarsanam V, Ray DK, et al Nitroimidazoles: Part XIX-Structure-activity Relationships. Indian J Chem 1964;23B:342-62.
- Nagarajan K, Shankar RG, Arya VP, George T, Nair MD, Shenoy SJ, et al. Nitroimidazoles; Part XXIII. Indian J ExpBiol 1992;30:193-200.
- Nagarajan K, Shankar RG, Rajappa S, Shenoy SJ. Nitroimidazoles XXI, 2,3-dihydro-6-nitroimidazo [2,1-b]oxazoles with anti-tubercular activity. Eur J Med Chem 1989;24:631-3.
- Ashtekar DR, Costa-Perira R, Nagrajan K, Vishvanathan N, Bhatt AD, Rittel W. In vitro and in vivo activities of the nitroimidazole CGI 17341 against Mycobacterium tuberculosis. Antimicrob Agents Chemother 1993;37:183-6.
- Masumoto M, Hashizume H, Tomishige T, Kawasaki M, Tsubouchi H. OPC-67683, a Nitro-Dihydro-Imidazooxazole derivative with promising action against tuberculosis in vitro and in mice. PLoS Med 2006;3:e466.
- Saliu OY, Crismale C, Schwander SK, Wallis RS. Bactericidal Activity of OPC-67683 against Drug-Tolerant Mycobacterium tuberculosis. J AntimicrobChemother 2007;60:994-8.
- Stover CK, Warrener P, VanDevanter DR, Sherman DR, Arain TM, Langhorne MH, et al. A small molecule nitroimidazopyran drug candidate for the treatment of tuberculosis. Nature 2000;405:962-6.
- Ginsberg AM, Laurenzi MW, Rouse DJ, Whitney KD, Spigelman MK. Safety, tolerability, and pharmacokinetics of PA-824 in healthy sunjects. Antimicrob Agents Chemother 2009;53:3720-5.
- Desjeux P. Leishmaniasis: Current situation and new perspectives. Comp ImmunolMicrobiol Infect Dis 2004;27:305-18.
- Available from: http://www.dndi.org/diseases-projects/diseases/vl.html, [Last accessed on 2013 Oct 6].
- Cunningham I. New culture medium for maintenance of tsetse tissues and growth of trypanosomatids. J Protozool 1977;24:325-9.
- Yang M, Arai C, Abu-Bakar MD, Lu J, Ge JF, Pudhom K, et al. Fluorinated Rhodacyanine (SJL-01) possessing high efficacy for Visceral Leishmaniasis (VL). J Med Chem 2010;53:368-73.
- Page B, Page M, Noel C. A new fluorometric assay for cytotoxicity measurements in vitro. J Oncol 1993;3;473-6.