- *Corresponding Author:
- Alaa Khedr
Department of Pharmaceutical Chemistry, King Abdulaziz University, Jeddah 21589, P.O. Box 80260, Saudi Arabia
E-mail: akhedr@kau.edu.sa
| Date of Submission | 24 February 2017 |
| Date of Revision | 18 October 2017 |
| Date of Acceptance | 27 May 2018 |
| Indian J Pharm Sci 2018;80(4):628-636 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
A gas chromatography-mass spectrometric method was optimized and applied to profile the major esterified fatty acids in eight lyophilized fish roe species and three marketed fish oil capsules. The method was able to separate efficiently 36 standard fatty acids as methyl esters using Teknokroma fused silica capillary column, 60 m×0.25 mm×0.2 μm. However, only 20 major esterified fatty acids were determined in fish roe and fish oil samples. The total amount of esterified fatty acids in salmon wild oil, salmon cold water oil and cod liver oil capsules were 235.5, 145.8, and 195.5 mg/g, respectively. However, the total amount of esterified fatty acids in fish roe species ranged from 40 to 61 mg/g. The quantitation data were treated with statistical principal component analysis, and showed a characteristic score value for each type of fish roe. Each type of fish roe showed a characteristic content profile of 20 major esterified fatty acids. The highest concentration of esterified omega-3, was found in salmon fish roe (15.34 mg/g) and Mosa fish roe (13.15 mg/g). The maximum amount of esterified α-linolenic acid, eicosapentaenoic acid and docosahexaenoic acid were found in salmon wild oil (7.73 mg/g), salmon fish roe (6.09 mg/g), and Mosa fish roe (11.00 mg/g), respectively. The developed method was found to be applicable for qualification and characterization of fish roe and fish oil.
Keywords
Fish roe, fish oil, esterified fatty acids, gas chromatography-mass spectrometry
The egg masses produced by the ovaries of different marine animals, is called fish roe (FR). The salted FR is called caviar and marketed as seafood. The FR is a rich source of omega-3 fatty acids [1]. Esposito et al. highlighted the impact of dietary factors in erectile dysfunction (ED) [2,3]. These authors concluded that adoption of healthy diets would help prevent ED. Shark liver has been shown as the richest source of phospholipids (PLs) and fatty acids [4]. Kiriakova et al. showed that essential PLs played a key role in the treatment of functional ED [5]. Furthermore, it has been reported that essential PLs enhance the activity of high-density lipoproteins to consume unconjugated cholesterol from the peripheral cells and to transport it in the esterified form to the liver and steroid producing glands [6]. Alvarez et al. showed the differential incorporation of fatty acids into and peroxidative loss of fatty acids from PLs of human spermatozoa [7]. Moreover, Hartree et al. studied the metabolism of plasmalogen and fatty acids in ramen semen [8]. A more interesting study showed the correlation between peroxidative breakdown of PLs in human spermatozoa, the spermicidal properties of fatty acid peroxides, and the protective action of seminal plasma [9]. Kaplan reported a detailed study about the hypoactive sexual desire and the common disorders of sexual desire [10]. He described the effect of dietary PLs and that esterified fatty acids (EFAs) could be a helpful supplement in ED. Kidd reported that omega-3 fatty acids including docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA) paly an essential role in the treatment of cognitive impairment and treatment of injured nerve [11]. The process of extraction of EFA and PL has been a big challenge. Lepage and Roy [12] optimized the conventional Folch’s extraction procedure [13] of EFA and improved the recovery to 95 % Moreover, Masood and Salem [14] showed a modified Lepage and Roy’s method and reported higher EFAs % recovery and better data precision. Han et al. [15] investigated the % recovery of total EFAs by spiking plasma with different concentrations of mono-stearin, as representative material, followed by extraction and transmethylation. The transmethylation procedure included sample treatment with 0.4 M potassium hydroxide in methanol, followed by neutralization with aqueous acetic acid and extraction with n-hexane. The reported recoveries from plasma were within the range of 83.1-91.2 %, with relative standard deviation values ranged from 9.4 to 10.3 %. To date, no reports have been found that described the extraction of EFA from FR. Numerous types of fish oil capsules are found in the market and labelled with the content of omega-3, regardless of the content of EFA and referred to as the most useful material. Most of these marketed supplementary gel capsules contain either fish oil or lyophilized FR. The type and quality of fish oil products is still ambiguous. The literature review revealed no analytical method that describe the qualification, full characterization, or profiling of active materials in fish oil or FR. Herein, we shall describe a validated analytical method for, separation, extraction, identification, and quantification of the most effective bioactive constituents.
The present investigation attempted to improve the extraction and develop a gas chromatography-mass spectrometric (GC-MS) method for the characterization of FR and fish oil and a species-based content profile of EFAs if any. The content of esterified α-linolenic acid (ALA), EPA and DHA were estimated in all studied FR species.
Materials and Methods
Wild Alaskan salmon oil (SALW), 1300 mg/soft gelatin capsule (lot: 711380, expiration: 12/2016; Natural Factors, Coquitlam, Canada), cold-water salmon oil (SALC) 1000 mg/soft gelatin capsule (lot: 415273-09, expiration: 03/2018; Nature’s Bounty Inc., Bohemia, NY, USA), Norwegian cod liver oil (COD), 415 mg/soft gelatin capsule (lot: 406717-06, expiration: 10/2016; Puritan’s Pride, Inc., Bohemia, NY, USA), fresh Alaskan salmon roe (SAL), golden (Bellevue, WA, USA), fresh Lump FR (LUM), red (Benfumat SA, Barcelona, Spain), and other fresh FR species ware purchased from fish market including; Hamour (HAM), Sevruga beluga (SEV), Tuna (TUN), Bory (BOR), Mosa (MOS), and Nagel (NAG). The fresh FR samples were freeze dried at –65° and kept in desiccator, under vacuum, for 7 d at –4° over CaO, powdered and stored at –85°, in tight closed glass containers. Supelco® 37 fatty acid methyl esters (FAME) standard mixture was purchased from Sigma Aldrich Co. (Saint Louis, MO, USA). The reference standard mix (1 ml) was transferred to screw-capped borosilicon mini-reaction vials 2 ml (v-shaped, with TFE liners, Alltech, GmbH, Unterhaching, Germany) and kept at –20°. The screw-capped (PTFE/silicon) 1 ml autosampler total recovery vials (12×32 mm) were purchased from Waters (Milford, MA, USA). Myristic acid ethyl ester and oleic acid ethyl ester were prepared in our laboratory, by acid esterification in H2SO4/ Et-OH. These esters were tested for purity by GC-MS (>99.6 %).
Chromatographic conditions
GC-MS, Clarus 500 GC/MS (PerkinElmer, Shelton, CT) was used. The software controller/integrator was TurboMass, version 5.4.2.1617 (PerkinElmer). Teknokroma TR-CN100 GC capillary column, (60 m×0.25 mm ID×0.20 μm df) was used (Teknokroma, Analitica SA, Barcelona, Spain). The carrier gas was helium (purity 99.9999 %) at a flow rate of 0.9 ml/min. Source (EI+) temperature, 210°; GC line temperature, 240°; electron energy, 70 eV; and trap emission, 100°. Oven program included initial temperature, 90° (hold 7 min), to 180° (rate 4.0°/min, hold 2.0 min), increased to 215° (rate 2.0°/min, hold 2 min); injector temperature, 225°; MS scan, 50 to 300 m/z; injection volume was 1.0 μl, splitless-split mode was applied (split ratio 5 % at 0 min, and 50 % at 0.2 min).
Extraction and trans-methylation
Adequate amount of dried FR was powdered in a porcelain mortar. A weight of 60 mg of this powder was transferred to glass test tube, mixed with 10 ml of CH3OH:CH2Cl2 (1:2, v/v), vortexed for 2 min, sonicated for 60 min at 45±2°, and centrifuged at 5000 rpm for 10 min. The clear supernatant was filtered using a syringe nylon membrane (0.2 μm). From this filtrate, a volume of 50 μl was transferred to 1.5-ml glass vial, dried with nitrogen gas at room temperature. The residue was reconstituted in 50 μl of toluene, mixed with 100 μl of 0.5 M potassium hydroxide/CH3OH, vortexed and the solution was maintained at 55° for 25 min using hot air oven). The vial was cooled and a volume of 250 μl water containing 20 % glacial acetic acid was added and vortexed for 10 s. The reaction mixture was mixed with the internal standard solution (25 μl) containing 50 ng/μl myristic acid ethyl ester as an internal standard and 50 ng/μl oleic acid ethyl ester as a control substance in n-hexane, vortexed and extracted with n-hexane (250 μl×3, 30 s). The n-hexane, supernatant layer, was withdrawn using a Pasteur pipette and transferred to a 1 ml total recovery vial leaving the aqueous layer (lower), dried with gentle stream of nitrogen gas at room temperature, the residue was reconstituted in 50 μl CH2Cl2 and a volume of 1.0 μl was injected for GC-MS analysis. Fish oil samples were prepared in toluene to get a concentration of 4 mg oil/ml, a volume of 50 μl was mixed with 100 μl 0.5 M potassium hydroxide/CH3OH and processed as described above.
Preparation of calibration solution
Microliter TLC syringes from Perkin Elmer were used for quantitative transfer of RS-Supelco® 37-FAME solution (Perkin Elmer Instruments, Australia, syringe with a replaceable leur-tip, 5, 10, and 100 μl capacity). The labeled concentrations of reference standard Supelco® 37 FAME solutions were 200, 400 or 600 ng/μl, as specified for each fatty acid in the leaflet. A stock solution was prepared by diluting 100 μl to 1 ml with CH2Cl2. This stock solution was used for preparation of 6 serial dilutions, spanning the range; 0.8 to 38.4 ng/μl of FAME with 25 ng/μl of internal standard and 25 ng/μl of control substance.
Extraction efficiency and method validation
Extraction efficiency was tested by spiking the FR samples with control fatty acid esters followed by extraction with MeOH:CH2Cl2 (1:2, v/v). Sixty milligrams of the dried-powdered FR was mixed with 10 ml MeOH:CH2Cl2 (1:2, v/v), spiked with 100 μl standard solutions containing ethyl ester of both myristic acid and oleic acid (control) at concentrations of 5, 50, 1000, and 5000 ng/ml, of each. The samples were extracted as described for FR, and a volume of 50 μl of the filtered extract was transferred to a 1.5 ml vial and dried with nitrogen gas. The residue was reconstituted in 50 μl toluene, mixed with 100 μl MeOH and vortexed. A volume of 250 μl water containing 20 % glacial acetic acid was added and vortexed for 10 s. This solution was extracted three times with 250 μl n-hexane. The supernatant was withdrawn using a Pasteur pipette and transferred to a total recovery 1-ml vial leaving the lower aqueous layer. The n-hexane layer was dried under a gentle stream of nitrogen gas at room temperature. The residue was reconstituted in 50 μl CH2Cl2 and a volume of 1.0 μl was injected for GC-MS analysis. The concentrations of control extracted were calculated from the calibration curve drawn by plotting the peak area versus concentration. The claimed concentration of the final injected standard solutions are 0.05, 0.5, 10.0 and 50.0 ng/μl. The fish oil samples were prepared in toluene and trans-methylated without prior extraction.
Analysis of EFA in FR and fish oil
Adequate amount of dried FR was powdered in a porcelain mortar. Sixty milligrams of this powder was transferred to a glass test tube, mixed with 10 ml of CH3OH:CH2Cl2 (1:2, v/v), vortexed for 2 min, sonicated for 60 min at 45±2°, and centrifuged at 5000 rpm for 10 min. The clear supernatant was filtered using a syringe nylon membrane (0.2 μm). From this filtrate, 50 μl was transferred to a 1.5 ml glass vial, dried under nitrogen gas at room temperature. The residue was reconstituted in 50 μl toluene, mixed with 100 μl 0.5 M potassium hydroxide/MeOH, vortexed, and the solution was maintained at 55° for 25 min (using a hot air oven). The vial was cooled and a volume of 250 μl water containing 20 % glacial acetic acid was added and vortexed for 10 s. The reaction mixture was mixed with 25 μl internal standard solution containing 50 ng/μl myristic acid ethyl ester as internal standard, and 50 ng/μl oleic acid ethyl ester as control substance, in n-hexane, vortexed and extracted with n-hexane (250 μl×3, 30 s). The n-hexane supernatant was withdrawn using a Pasteur pipette and transferred to total recovery 1 ml vial leaving out the lower aqueous layer, dried under a gentle stream of nitrogen gas at room temperature, the residue was reconstituted in 50 μl CH2Cl2 and a volume of 1.0 μl was injected for GC-MS analysis. Fish oil samples were prepared in toluene to get a concentration of 4 mg oil/ml, a volume of 50 μl was mixed with 100 μl 0.5 M potassium hydroxide/MeOH and processed as described above. The principle component analysis was calculated by using a software Multivariate Statistical Package, version 3.22, Copyrights 1985-2013, Kovach computing services.
Results and Discussion
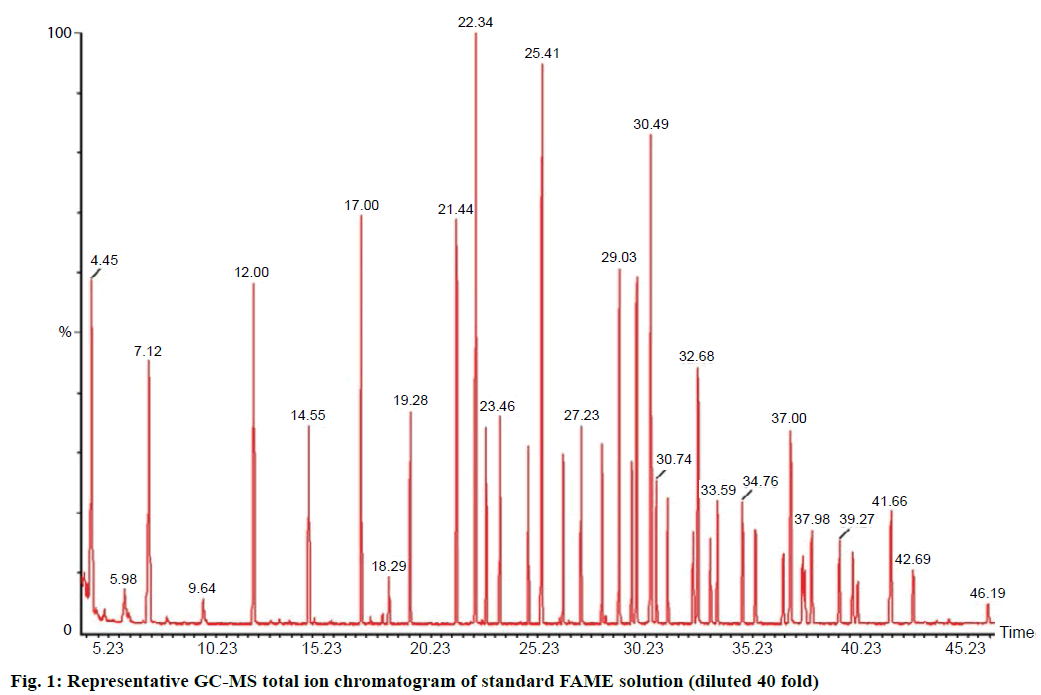
Thirty-six fatty acids as methyl esters were successfully separated and detected using Teknokroma GC column (60 m) applying the temperature program mentioned in the experimental part (Figure 1). During sample preparation it was noticed that EFA tended to volatilize upon exposure to nitrogen gas at 40°, especially short chain fatty acids (C4 to C14). Minimization of sample vaporization during injection was achieved when the injector temperature adjusted to 225° and applying splitless vent mode for short time period at the injection time. These adjustments allowed minimal venting of EFA from GC injector and precise accumulation of analyte travelled with carrier gas to the column. Also, enough temperature equilibration time, 5 min, showed precise retention times. Moreover, the most critical step in sample preparation was the drying of solvent containing FAMEs. Table 1 showed the chromatographic performance parameters of standard FAME solution mixture. Only three FAMEs that showed USP resolution value less than 1.5, including C18:1n9c, C20:4n6, and C20:5n3. However, the standard deviation of the peak area, of the three compounds, was not more than 2.88. The software was programmed to extract the average total ion mass spectrum and accurate full integration of the three overlapped peaks. The major 20 EFAs found in FR and fish oil were characterized by matching the retention time, mass spectrum of standard mixture, and NIST2008 database.
| FAME | tRa, min | Concentration, ng μl-1 | (W0.05)a, S | ka | αa | USP resolution | USP plate count |
|---|---|---|---|---|---|---|---|
| C6:0 | 4.45 | 10.0 | 0.13 | 1.78 | 570 | ||
| C8:0 | 7.12 | 10.0 | 0.13 | 3.45 | 1.94 | 21.36 | 911 |
| C10:0 | 12.00 | 10.0 | 0.20 | 6.50 | 1.88 | 30.03 | 960 |
| C11:0 | 14.55 | 5.0 | 0.14 | 8.09 | 1.25 | 15.00 | 1663 |
| C12:0 | 17.00 | 10.0 | 0.13 | 9.63 | 1.19 | 18.15 | 2092 |
| C13:0 | 19.28 | 5.0 | 0.11 | 11.05 | 1.15 | 19.00 | 2804 |
| C14:0 | 21.44 | 10.0 | 0.12 | 12.40 | 1.12 | 18.78 | 2859 |
| InSt | 22.34 | 25.0 | 0.14 | 12.96 | 1.05 | 6.92 | 2553 |
| C14:1 | 22.80 | 5.0 | 0.11 | 13.25 | 1.02 | 3.68 | 3316 |
| C15:0 | 23.46 | 5.0 | 0.11 | 13.66 | 1.03 | 6.00 | 3412 |
| C15:1 | 24.78 | 5.0 | 0.12 | 14.49 | 1.06 | 11.48 | 3304 |
| C16:0 | 25.41 | 15.0 | 0.13 | 14.88 | 1.03 | 5.04 | 3127 |
| C16:1 | 26.40 | 5.0 | 0.12 | 15.50 | 1.04 | 7.92 | 3520 |
| C17:0 | 27.23 | 5.0 | 0.10 | 16.02 | 1.03 | 7.55 | 4357 |
| C17:1 | 28.20 | 5.0 | 0.12 | 16.63 | 1.04 | 8.82 | 3760 |
| C18:0 | 29.03 | 10.0 | 0.11 | 17.14 | 1.03 | 7.22 | 4223 |
| C18:1n9t | 29.65 | 5.0 | 0.13 | 17.53 | 1.02 | 5.17 | 3649 |
| C18:1n9c | 29.82 | 10.0 | 0.14 | 17.64 | 1.01 | 1.26 | 3408 |
| CTRL | 30.49 | 25.0 | 0.16 | 18.06 | 1.02 | 4.47 | 3049 |
| C18:2n6t | 30.74 | 5.0 | 0.13 | 18.21 | 1.01 | 1.72 | 3783 |
| C18:2n6c | 31.27 | 5.0 | 0.14 | 18.54 | 1.02 | 3.93 | 3574 |
| C18:3n6 | 32.45 | 5.0 | 0.14 | 19.28 | 1.04 | 8.43 | 3709 |
| C20:0 | 32.68 | 10.0 | 0.17 | 19.43 | 1.01 | 1.48 | 3076 |
| C18:3n3 | 33.25 | 5.0 | 0.15 | 19.78 | 1.02 | 3.56 | 3547 |
| C20:1 | 33.59 | 5.0 | 0.15 | 19.99 | 1.01 | 2.27 | 3583 |
| C21:0 | 34.76 | 5.0 | 0.14 | 20.73 | 1.04 | 8.07 | 3973 |
| C20:2 | 35.35 | 5.0 | 0.14 | 21.09 | 1.02 | 4.21 | 4040 |
| C20:3n6 | 36.64 | 5.0 | 0.14 | 21.90 | 1.04 | 9.21 | 4187 |
| C22:0 | 37.00 | 10.0 | 0.20 | 22.13 | 1.01 | 2.12 | 2960 |
| C20:3n3 | 37.56 | 5.0 | 0.13 | 22.48 | 1.02 | 3.39 | 4623 |
| C20:4n6 | 37.66 | 5.0 | 0.12 | 22.54 | 1.00 | 0.80 | 5021 |
| C22:1n9 | 37.98 | 5.0 | 0.19 | 22.74 | 1.01 | 2.06 | 3198 |
| C23:0 | 39.27 | 5.0 | 0.18 | 23.54 | 1.04 | 6.97 | 3491 |
| C22:2 | 39.87 | 5.0 | 0.18 | 23.92 | 1.02 | 3.33 | 3544 |
| C20:5n3 | 40.10 | 5.0 | 0.16 | 24.06 | 1.01 | 1.35 | 4010 |
| C24:0 | 41.66 | 10.0 | 0.21 | 25.04 | 1.04 | 8.43 | 3174 |
| C24:1 | 42.69 | 5.0 | 0.22 | 25.68 | 1.03 | 4.79 | 3105 |
| C22:6n3 | 46.19 | 5.0 | 0.22 | 27.87 | 1.09 | 15.91 | 3359 |
atR, retention time; W0.05, peak width at 95 % height from peak apex; k, retention factor; and α, selectivity coefficient
Table 1: Chromatographic performance parameters of standard 36 FAME solution mixtures
The commonly applied method previously reported by Folch et al. for the extraction and trans-methylation of EFAs, was modified and validated to quantitate EFA in different species of FR and fish oils [13]. Fish oil samples were prepared in toluene followed by trans-methylation without prior extraction. However, the differences in size and shape of FR samples needed special treatment to confirm efficient extraction of lipid content. Fresh FR samples were freeze dried and kept 7 d over anhydrous CaO before powdered in a porcelain mortar to fine particles in a homogenous form. The percent error due to sample drying with nitrogen gas was measured by matching the peak area responses of FAME standard mixture, diluted 40 folds, with the same concentration dried with nitrogen gas and diluted. The short chain FAME, including C4–C8, showed high percent error equal to 3.77 due to partial evaporation of the standard materials with solvent, even at room temperature. The final n-hexane extract was spiked with myristic acid ethyl ester as internal standard and oleic acid ethyl ester as control material. The percent loss of FAME due to evaporation with nitrogen gas was calculated from the relative peak area of control monitored in sample to that in standard solution mix.
The extraction recovery was tested by spiking 60 mg of salmon FR (Wild Alaskan) with standard solution mixture containing myristic acid ethyl ester and oleic acid ethyl ester, followed by GC-MS analysis. Four concentration levels, including the lower limit of quantification (LLOQ), were used (Table 2). The concentrations of internal standard and oleic acid ethyl ester were calculated from the corresponding calibration curve of each drawn by plotting the peak area versus concentration in ng/μl. In the present study, both myristic acid ethyl ester and oleic acid ethyl ester were used as calibration materials, neither as internal standard nor control material. The percent recovery of both standard substances was within the range of 97.9 to 100.7 %. The collected FR sample was different in size (egg size) and shell thickness, and showed fluctuations in recovery upon extraction without prior grinding to fine particles. Sonication of the dried and powdered FR suspended in MeOH:CH2Cl2 (1:2, v/v) at 45±2° showed a precise percent recovery with RSD ranging from 2.81 to 0.10 and percent relative error ranging from 0.71 to –2.09.
| Concentration, μg/g | Myristic acid ethyl ester | Oleic acid ethyl ester | ||
|---|---|---|---|---|
| Mean recovery (%)±SDa | Er (%)b | Mean recovery (%)±SDa | Er (%)b | |
| 8.33 (LLOQ) | 98.66±0.34 | –1.34 | 97.91±2.81 | –2.09 |
| 83.33 | 99.80±0.14 | –0.20 | 98.92±0.40 | –1.08 |
| 1666.66 | 99.33±0.15 | –0.67 | 98.93.22±0.77 | –1.07 |
| 8333.33 | 100.04±0.10 | 0.04 | 100.71±0.76 | 0.71 |
aMean recovery (%)±RSD of three determinations; bpercent relative error
Table 2: Percent recovery of myristic acid ethyl ester and oleic acid ethyl ester from spiked salmon fish roe
The peak area ratios of each standard FAME to internal standard were found linear in the concentration range listed in Table 3. The regression coefficients were close to unity. The limit of detection (LOD) and LOQ of each FAME was calculated as concentration level that gives GC-MS response of at least three or nine times the base line noise, respectively (Table 3). The concentrations of FAME in samples were calculated from the corresponding calibration curve taking in account the percent recovery of each compound. For testing the method precision and accuracy, oleic acid ethyl ester was used as representative FAME material. Two representative matrices were used including LUM and SALW. FR samples were spiked with specified concentrations of control, extracted and trans-methylated. However, the fish oil matrix was dissolved in toluene; trans-methylated, spiked with specified concentration of control and analysed by GC-MS. Table 4 summarized the intraday and interday precision and accuracy of LLOQ, low, medium and high concentration levels. The results were within the acceptable range.
| Calibration curves | ||||||
|---|---|---|---|---|---|---|
| EFA | Slope (6 levels) |
Intercept | r2 | Working range, ng/μl | LOQ, ng/ml | LOD, ng/ml |
| C10:0 | 0.09446 | –0.02035 | 0.9973 | 1.6-25.6 | 42 | 14 |
| C14:0 | 0.08102 | –0.02173 | 0.9994 | 1.6-25.6 | 48 | 16 |
| C15:0 | 0.07812 | –0.01295 | 0.9992 | 0.8-12.8 | 54 | 18 |
| C16:0 | 0.08137 | –0.03413 | 0.9994 | 2.4-38.4 | 48 | 16 |
| C16:1 | 0.06870 | –0.01117 | 0.9993 | 0.8-12.8 | 66 | 22 |
| C17:0 | 0.07558 | –0.01113 | 0.9996 | 0.8-12.8 | 60 | 20 |
| C18:0 | 0.07675 | –0.02328 | 0.9993 | 1.6-25.6 | 54 | 18 |
| C18:1n9c | 0.07781 | –0.02664 | 0.9991 | 1.6-25.6 | 54 | 18 |
| C18:2n6t | 0.06112 | –0.00940 | 0.9993 | 0.8-12.8 | 72 | 24 |
| C18:2n6c | 0.06018 | –0.02002 | 0.9971 | 0.8-12.8 | 72 | 24 |
| C18:3n6 | 0.04835 | –0.00756 | 0.9991 | 0.8-12.8 | 90 | 30 |
| C20:0 | 0.07458 | –0.02105 | 0.9993 | 1.6-25.6 | 66 | 22 |
| C18:3n3 | 0.04678 | –0.00767 | 0.9991 | 0.8-12.8 | 90 | 30 |
| C20:1 | 0.07049 | –0.00780 | 0.9985 | 0.8-12.8 | 66 | 22 |
| C20:2 | 0.05578 | –0.00853 | 0.9989 | 0.8-12.8 | 72 | 24 |
| C20:4n6 | 0.01250 | 0.00518 | 0.9856 | 0.8-12.8 | 240 | 80 |
| C22:1n9 | 0.06421 | –0.00919 | 0.9989 | 0.8-12.8 | 72 | 24 |
| C20:5n3 | 0.02929 | –0.00304 | 0.9992 | 0.8-12.8 | 120 | 40 |
| C24:1 | 0.05454 | –0.00811 | 0.9979 | 0.8-12.8 | 90 | 30 |
| C22:6n3 | 0.01669 | –0.00017 | 0.9915 | 0.8-12.8 | 180 | 60 |
Table 3: Calibration parameters of the standard mixture of fame solution
| Concentration, μg/g | Lump FR | SALC oil | ||||||
|---|---|---|---|---|---|---|---|---|
| Intraday | Interday | Intraday | Interday | |||||
| Mean recover (%)±SDa | Er (%)b | Mean recover (%)±SDa | Er (%)b | Mean recover (%)±SDa | Er (%)b | Mean recover (%) ±SDa | Er (%)b | |
| 8.33 (LLOQ) | 97.95±1.66 | –2.05 | 98.18±2.11 | –1.82 | 99.77±0.80 | –0.23 | 99.68±1.60 | –0.32 |
| 83.33 | 99.05±1.68 | –0.95 | 98.97±2.05 | –1.03 | 97.89±1.50 | –2.11 | 97.82±0.88 | –2.18 |
| 1666.66 | 98.92±0.25 | –1.08 | 96.66±0.10 | –3.34 | 99.98±0.11 | –0.02 | 99.99±0.08 | –0.01 |
| 8333.33 | 101.05±0.05 | 1.05 | 99.15±0.03 | –0.85 | 99.89±0.07 | –0.11 | 100.01±0.02 | 0.01 |
aMean recovery (%) ± SD of three determinations; bpercent relative error
Table 4: Evaluation of the intraday and interday accuracy and precision for the determination of spiked lump FR and SALC oil with oleic acid ethyl ester
Sample stability was tested by analysis of eight different FR samples kept in refrigerator for 30 d at –20°. The relative standard deviation of all EFA in these reserved samples was not more than 0.55 %. The GC-MS run time was 50 min, plus 2 min equilibration time. The sample ready for injection, containing 25 ng/μl control, was analysed at zero, 12, and 24 h. The autosampler stability testing showed a % RSD of not more than 0.66, however, samples left at room temperature for more than 36 h showed % RSD equal to 3.25 %.
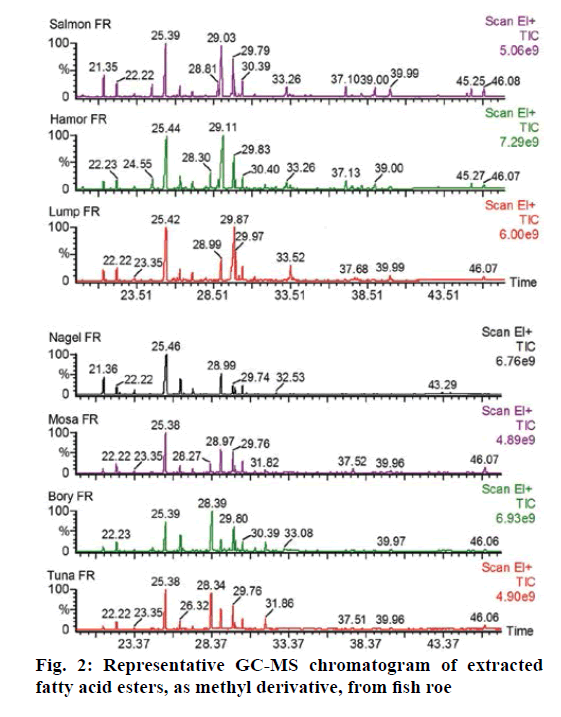
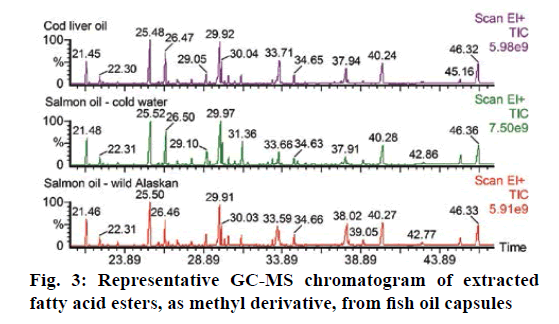
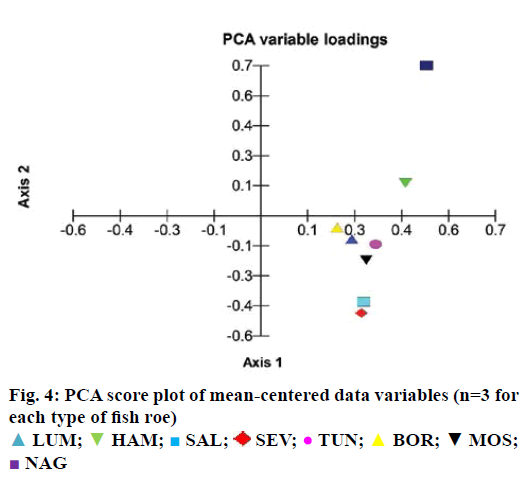
The GC-MS data showed 20 major EFA in FR and fish oil. Oleic acid ethyl ester was spiked to trans-methylated residue at a concentration of 25 ng/μl before the step of drying with nitrogen gas to calculate the percent loss before considering the results. Twenty peaks were defined to the TurboMass software by retention time and average mass spectrum. The samples containing high concentrations were diluted with CH2Cl2 and reinjected, to obtain a response within the calibration range. Figures 2 and 3 showed the representative GC-MS chromatogram of eight FR samples and three fish oil samples. The quantitative data showed that fish oil samples contain higher concentration of total EFA than FR (Table 5). Moreover, the content profiles of EFA could characterize the sample analysed. The quantified EFAs in each sample were statistically analysed using principal component analysis (PCA). As shown in Figure 4, the EFAs in each sample have distinctly different EFA score from other species. The PCA calculated as a function of variances of EFAs content per each FR species.
| Concentration of EFA, mg/g* | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| EFA | LUM | HAM | SAL | SEV | TUN | BOR | MOS | NAG | SALW | SALC | CODL |
| C10:0 | 0.08 | 0.04 | 0.04 | 0.08 | 0.10 | 0.08 | 0.10 | 0.10 | 0.35 | 0.20 | 0.17 |
| C14:0 | 1.12 | 1.52 | 3.23 | 0.93 | 0.89 | 1.12 | 0.74 | 5.78 | 18.78 | 24.59 | 19.59 |
| C15:0 | 0.28 | 0.62 | 0.43 | 0.14 | 0.34 | 0.42 | 0.35 | 1.26 | 2.36 | 1.68 | 1.58 |
| C16:0 | 13.96 | 24.72 | 13.26 | 13.43 | 17.52 | 11.77 | 15.49 | 30.36 | 52.95 | 62.50 | 50.14 |
| C16:1 | 1.79 | 3.57 | 1.88 | 0.71 | 2.90 | 5.45 | 2.08 | 5.68 | 22.95 | 1.32 | 1.54 |
| C17:0 | 0.77 | 2.04 | 0.85 | 0.26 | 1.00 | 0.55 | 0.99 | 2.03 | 4.77 | 2.52 | 3.89 |
| C18:0 | 5.18 | 2.12 | 14.92 | 9.59 | 7.21 | 3.55 | 7.75 | 9.58 | 10.39 | 0.21 | 10.19 |
| C18:1n9c | 9.82 | 9.78 | 7.10 | 15.44 | 7.74 | 8.10 | 5.93 | 2.90 | 38.63 | 0.00 | 51.95 |
| C18:2n6t | 0.06 | 0.08 | 0.03 | 0.06 | 0.17 | 0.05 | 0.05 | 0.04 | 1.14 | 0.20 | 2.82 |
| C18:2n6c | 0.30 | 0.35 | 0.07 | 6.50 | 0.64 | 1.30 | 0.07 | 0.19 | 9.66 | 32.97 | 10.35 |
| C18:3n6 | 0.04 | 0.13 | 0.05 | 0.05 | 0.07 | 0.16 | 0.08 | 0.07 | 1.99 | 0.09 | 0.00 |
| C20:0 | 0.10 | 0.41 | 0.18 | 0.48 | 0.20 | 0.14 | 0.15 | 0.48 | 0.78 | 0.78 | 0.57 |
| C18:3n3 (ALA) | 0.05 | 0.27 | 0.03 | 0.03 | 1.24 | 1.24 | 0.29 | 0.11 | 7.73 | 3.27 | 5.99 |
| C20:1 | 2.50 | 1.15 | 0.44 | 0.71 | 0.16 | 0.36 | 0.19 | 0.36 | 16.86 | 13.51 | 0.00 |
| C20:2 | 0.07 | 0.15 | 0.47 | 0.09 | 0.26 | 0.17 | 0.11 | 0.07 | 0.07 | 0.07 | 1.60 |
| C20:4n6 | 3.50 | 6.21 | 1.09 | 1.75 | 4.09 | 1.05 | 7.74 | 0.35 | 8.12 | 0.00 | 10.90 |
| C22:1n9 | 0.41 | 0.09 | 0.06 | 0.04 | 0.06 | 0.04 | 0.05 | 0.05 | 37.01 | 1.47 | 23.84 |
| C20:5n3 (EPA) | 1.53 | 2.44 | 6.09 | 0.31 | 1.52 | 1.69 | 1.86 | 0.18 | 0.43 | 0.31 | 0.18 |
| C24:1 | 0.11 | 0.13 | 0.03 | 0.10 | 0.14 | 0.03 | 0.34 | 0.34 | 0.52 | 0.09 | 0.21 |
| C22:6n3 (DHA) | 1.92 | 7.94 | 9.22 | 0.24 | 9.49 | 3.64 | 11.00 | 0.42 | 0.03 | 0.01 | 0.03 |
| Total omega-3 | 3.50 | 10.65 | 15.34 | 0.58 | 12.25 | 6.57 | 13.15 | 0.71 | 8.19 | 3.59 | 6.20 |
| Unsaturated EFA | 22.10 | 32.27 | 26.56 | 26.01 | 28.47 | 23.27 | 29.78 | 10.76 | 145.14 | 53.31 | 109.41 |
| Saturated EFA | 21.49 | 31.48 | 32.90 | 24.92 | 27.27 | 17.64 | 25.58 | 49.60 | 90.38 | 92.48 | 86.13 |
| Total EFA | 43.59 | 63.75 | 59.46 | 50.93 | 55.74 | 40.91 | 55.36 | 60.36 | 235.52 | 145.79 | 195.53 |
*The percent of relative standard deviation of all determined concentrations was not more than 0.25 %
Table 5: concentration of EFA in eight species of fish roe and three types of marketed fish oil capsules
The highest concentration of esterified omega-3, was found in salmon FR (15.34 mg/g) and MOS FR (13.15 mg/g). The maximum amounts of esterified ALA, EPA and DHA were found in SALW oil (7.73 mg/g), SAL FR (6.09 mg/g), and MOS FR (11.00 mg/g), respectively. Generally, fish oils are rich in esterified ALA than FR. Also, FR is rich in DHA than fish oil (Table 5). The total amount of esterified EFAs was relatively high in salmon oil, 145.14 mg/g. The results revealed that fish oils contain double the amount of EFAs compared to FR. Most marketed fish oil capsules labelled the total omega-3 content ignoring the concentration of EFAs.
An efficient GC-MS quantification method for EFAs in FR and fish oils was optimized and validated. The extraction recovery of EFA from FR and fish oil was monitored during analysis by using oleic acid ethyl ester. The optimized GC-MS method was capable to discriminate and characterize the species of FR and fish oil based on the content of EFAs, and esterified omega-3. The total amount of esterified omega-3, including ALA, EPA and DHA were high in fish oil compared with FR samples. Salmon FR followed by MOS, are rich in esterified EFA. Each species of FR or fish oil have showed a characteristic profile based on the content of EFAs.
Acknowledgements
This project was funded by the Deanship of Scientific Research (DSR), at King Abdulaziz University, Jeddah, under grant no. 147/166/1437. The authors, therefore, acknowledge with thanks DSR for technical and financial support.
Conflicts of interest
The authors declare no competing interests.
References
- Rincón-Cervera MÁ, Suárez-Medina MD, Guil-Guerrero JL. Fatty acid composition of selected roes from some marine species. Eur J Lipid Sci Technol 2009;111:920-5.
- Esposito K, Ciotola M, Giugliano F, De Sio M, Giugliano G, D'armiento M, et al. Mediterranean diet improves erectile function in subjects with the metabolic syndrome. Int J Impot Res 2006;18:405-10.
- Esposito K, Giugliano F, Maiorino MI, Giugliano D. Dietary factors, Mediterranean diet and erectile dysfunction. J Sex Med 2010;7(7):2338-45.
- Chen S, Li KW. Mass spectrometric identification of molecular species of phosphatidylcholine and lysophosphatidylcholine extracted from shark liver. J Agric Food Chem 2007;55(23):9670-7.
- Kiriakova N, Kiriakov A, Schneider E, Bonev A. Therapeutic effect of essential phospholipids on functional sexual disorders in males. J Eur Acad Dermatol Venerol 1998;11(2):191-3.
- Johnson WJ, Bamberger MJ, Latta RA, Rapp PE, Phillips MC, Rothblat GH. The bidirectional flux of cholesterol between cells and lipoproteins. Effects of phospholipid depletion of high density lipoprotein. J Biol Chem 1986;261(13):5766-76.
- Alvarez JG, Storey BT. Differential incorporation of fatty acids into and peroxidative loss of fatty acids from phospholipids of human spermatozoa. Mol Reprod Dev 1995;42(3):334-46.
- Hartree EF, Mann T. Phospholipids in ram semen: metabolism of plasmalogen and fatty acids. Biochem J 1961;80(3):464-76.
- Jones R, Mann T, Sherins R. Peroxidative breakdown of phospholipids in human spermatozoa, spermicidal properties of fatty acid peroxides, and protective action of seminal plasma. Fertil Steril 1979;31(5):531-7.
- Kaplan HS. Hypoactive sexual desire. J Sex Marital Ther 1977;3(1):3-9.
- Kidd PM. Omega-3 DHA and EPA for cognition, behavior, and mood: clinical findings and structural-functional synergies with cell membrane phospholipids. Altern Med Rev 2007;12(3):207-27.
- Lepage G, Roy CC. Direct transesterification of all classes of lipids in a one-step reaction. J Lipid Res 1986;27(1):114-20.
- Folch J, Lees M, Sloane Stanley GH. A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 1957;226:497-509.
- Masood MA, Salem N Jr. High-throughput analysis of plasma fatty acid methyl esters employing robotic transesterification and fast gas chromatography. Lipids 2008;43(2):171-80.
- Han LD, Xia JF, Liang QL, Wang Y, Wang YM, Hu P, et al. Plasma esterified and non-esterified fatty acids metabolic profiling using gas chromatography-mass spectrometry and its application in the study of diabetic mellitus and diabetic nephropathy. Anal Chim Acta 2011;689(1):85-91.
 LUM;
LUM;  HAM;
HAM;  SAL;
SAL;  SEV;
SEV;  TUN;
TUN;  BOR;
BOR;  MOS;
MOS;  NAG
NAG



