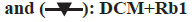- *Corresponding Author:
- F. Zhang
Department of Cardiology, Jinhua People’s Hospital, Jinhua, Zhejiang province 321000, China
E-mail: zhangfen2021@163.com
| Date of Received | 25 June 2021 |
| Date of Revision | 10 August 2021 |
| Date of Acceptance | 08 June 2022 |
| Indian J Pharm Sci 2022;84(3):723-729 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
Ginsenosides play an important role in the treatment of diabetes and obesity. Our group predicted that ginsenoside Rb1 combined with insulin would have a protective effect in diabetic cardiomyopathy. Diabetic model was established by high fat feeding combined with intraperitoneal injection of 70 mg/ kg streptozotocin in 8 w old male Sprague-Dawley rats. One rat was sacrificed at 8 w, 12 w and 14 w, respectively. The cardiac function and pathological structure of the myocardium were observed. It was concluded that the model of the diabetic cardiomyopathy group was successful. Rats were randomly divided into 4 groups and treated with different doses for 6 w. During this period, all rats received regular blood from the tail vein, had regular random blood glucose measurements and had regular fasting body weight measurements. After 6 w of intervention, cardiac ultrasonography was performed to evaluate cardiac function in all groups. Blood samples of aorta were collected for biochemical detection and pathological staining, immunohistochemical staining, reverse transcription polymerase chain reaction, Western blot and enzyme-linked immunosorbent assay were taken from heart tissue samples for detection. Compared with the normal group, diabetic cardiomyopathy rats displayed severe hyperglycemia, lower body weight, poorer cardiac performance, cardiac fibrosis and cardiac inflammation. Diabetic cardiomyopathy rats suffered obvious effects from over-activation of the heparin-binding epidermal growth factor pathway. Ginsenoside Rb1 treatment attenuated hyperglycemia, body weight loss and cardiac injury as well as heparin-binding epidermal growth factor activation induced by diabetic cardiomyopathy. Meanwhile, ginsenoside Rb1 had no obvious toxicity or side effects in the heart, liver or kidneys. Ginsenoside Rb1 treatment in diabetic rats can inhibit the over-activated heparin-binding epidermal growth factor signaling pathway, attenuate fibrosis of myocardial tissue and improve cardiac function, and has no obvious toxicity or side effects. It is a potential drug for diabetic heart disease.
Keywords
Ginsenoside Rb1, diabetic cardiomyopathy, heparin-binding epidermal growth factor pathway, atherosclerosis, hypertension
Diabetes is a disease with one of the highest incidence rates and death tolls in the world, affecting approximately 422 million patients. By 2050, about 30 % of the United States of America (USA) population will most likely suffer from diabetes[1]. Diabetes has gradually become a major factor in the national economy and everyday livelihoods in China, with nearly 10.9 % morbidity in 2013[2]. Long-term diabetic complications are considered to be largely the result of vascular injury and can be divided into macrovascular and microvascular complications[3]. 90 % of myocardial blood supply comes from microvessels and the remaining 10 % comes from the large blood vessels on the epicardial surface. Diabetic Cardiomyopathy (DCM) is defined as ventricular dysfunction without coronary atherosclerosis and hypertension[4]. The pathogenesis of DCM involves multiple mechanisms, including metabolic and inflammatory imbalances, structural myocardial changes (fibrosis), peripheral neuropathy and coronary microvascular dysfunction[5]. As one of the most common complications of diabetes, DCM treatment is of great significance to prevent diabetes-related mortality.
According to commonly accepted medical guidelines, lifestyle interventions such as diet and exercise are the foundation of diabetes treatment. If lifestyle interventions fail, medication should be prescribed. Oral drugs, insulin and insulin analogues are used to treat Type 2 Diabetes (T2D). Some Chinese medicines and their natural active ingredients have a hypoglycemic effect. Ginseng[6] is commonly used to treat hemoptysis, hemostasis and hematoma in Chinese traditional medicine. Ginsenoside Rb1 (the main extract of ginseng) has the effect of improving insulin sensitivity and may be used as an anti-diabetic drug. As reported, ginseng and ginsenoside Rb1 can inhibit weight gain[7], lower blood glucose[8,9] and improve insulin sensitivity[10], results which may be achieved by improving glucose tolerance in diabetic patients, reducing liver fat accumulation, inhibiting lipolysis of adipocytes[11] and regulating the development and function of adipocytes[9,12,13]. These effects are very beneficial for the treatment of diabetes and its complications.
Following the literature, we speculated that ginsenoside Rb1 could play an important role in treating DCM. This study mainly explored the role of ginsenoside Rb1 and its molecular mechanism in DCM.
Materials and Methods
Animal model and treatment:
Forty 8 w old male Sprague-Dawley (SD) rats were randomly divided into two groups. The experimental group was fed a high-fat diet for 6 w and the diabetic model was induced by a single intraperitoneal injection of streptin (Streptozotocin (STZ), 30 mg/kg, dissolved in saline). Random blood glucose was measured using a glucose meter (Roche, German). Rats with blood glucose level >16.8 mmol/l were selected in DCM group. It was diagnosed as T2D. If the blood glucose is not up to the standard, the peripheral blood glucose will be detected twice within 1 w. If the blood glucose is still <16.8 mmol/l, the blood glucose will be kicked out. Experimental group continues with high fat feed, respectively in 8 w, 12 w and 14 w executed two rats, observe the cardiac function and myocardial pathologic structure, in w 12 echocardiographic indicates cardiac function decline, pathological myocardial fibrosis, 14 w once again confirmed, that a DCM group building success. The control group was intraperitoneally injected with citrate buffer solution at 30 mg/kg. The control group was fed with ordinary feed all the time.
Next, the DCM group was randomly divided into two groups; one group was treated with insulin only and the other group was treated with insulin and ginsenoside Rb1. The normal group was randomly divided into normal saline group and ginsenoside Rb1 group, with 6 cases in each group. Rats were kept in laminar flow cages where they were subjected to 12 h/12 h dark/ light cycles and had free access to standard food and running water. 6 w later, we measured cardiac function. The heart tissue and blood samples of the rats were sacrificed for histological and molecular analysis. The research was approved by the Institutional Animal Care Use Committee of Jinhua People's Hospital.
Cardiac function measurement:
Echocardiography was carried out using a Vivid 7 ultrasound system with a 10-MHz transducer (General Electric, USA). Rats were anesthetized by inhalation of 2 % isoflurane in oxygen, Left Ventricular End-Diastolic Diameter (LVEDD) and Left Ventricular End-Systolic Diameter (LVESD) were measured on the parasternal left ventricular long-axis view. These chamber dimensions were indexed to body weight. Left Ventricular Ejection Fraction (LVEF) and Fractional Shortening (FS) were calculated by assuming a spherical left ventricular geometry for the ultrasound system algorithms. The data above was measured at least three times and averaged. All measurements were operated by an experienced investigator who was blinded to the grouping.
Histopathology:
Heart samples were fixed by formalin and embedded in paraffin. For Hematoxylin and Eosin (HE) staining, the 3 μm tissue sections were deparaffinized and stained with HE stain. For Masson staining, the heart sections were deparaffinized and incubated with 100 μl of Masson staining solution for 5 min. Then the sections were washed with distilled water, incubated with 100 μl of phosphomolybdic acid reagent for 5 min and incubated with 100 μl of aniline blue for 5 min. After treatment with 100 μl of differentiation solution for 30-60 s, sections were dehydrated with gradient ethanol, made transparent with xylene and then sealed.
Human Heparin-Binging Epidermal Growth Factor (HB-EGF) Enzyme-Linked Immunosorbent Assay (ELISA):
A Human HB-EGF ELISA kit (Elabscience, China) was used to test for the activity of HB-EGF in plasma, according to the manufacturer’s instructions.
Ribonucleic Acid (RNA) isolation and Reverse Transcription quantitative Polymerase Chain Reaction (RT-qPCR):
Total RNA was extracted by TRIzol reagent (Invitrogen, USA) and reverse-transcribed using a PrimeScript RT reagent kit with genomic Deoxyribonucleic Acid (gDNA) eraser (Takara, Japan) according to the manufacturer’s instructions. In addition, PCR was carried out with FastStart™ Universal SYBR Green Master (ROX) (Roche, German) using the ABI 7900 System (Applied Biosystems, USA) according to the manufacturer's instructions. Beta (β)-actin was chosen as an internal control. The primers used for RT-qPCR are Interleukin-6 (IL-6) forward: CTGCAAGAGACTTCCATCCAG; reverse: AGTGGTATAGACAGGTCTGTTGG; β-actin forward: GTTGGAGCAAACATCCCCCA and reverse: ACGCGACCATCCTCCTCTTA.
Western blot:
Samples were disintegrated by Radioimmunoprecipitation Assay (RIPA) buffer and quantified with a Bicinchoninic Acid (BCA) assay kit (Pierce, USA). Total protein was isolated within sodium dodecyl-sulfate polyacrylamide gel by electrophoresis after denaturation and transferred to polyvinylidene fluoride membranes. The bands were blocked using 5 % non-fat milk for 1 h and then incubated with primary antibodies overnight. Then, the bands were incubated with secondary antibodies. Proteins were detected using an Enhanced Chemiluminescence (ECL) kit (CST, USA). The HB-EGF antibody used for Western blot was purchased from Abcam (UK).
Statistical analysis:
Statistical analysis was conducted using Statistical Package for the Social Sciences (SPSS) 20.0 for Windows (SPSS Inc., USA). Continuous variables were expressed as the mean±standard deviation. Categorical variables were analyzed by the Chi-square (χ2) test and continuous variables were analyzed by the t-test.
Results and Discussion
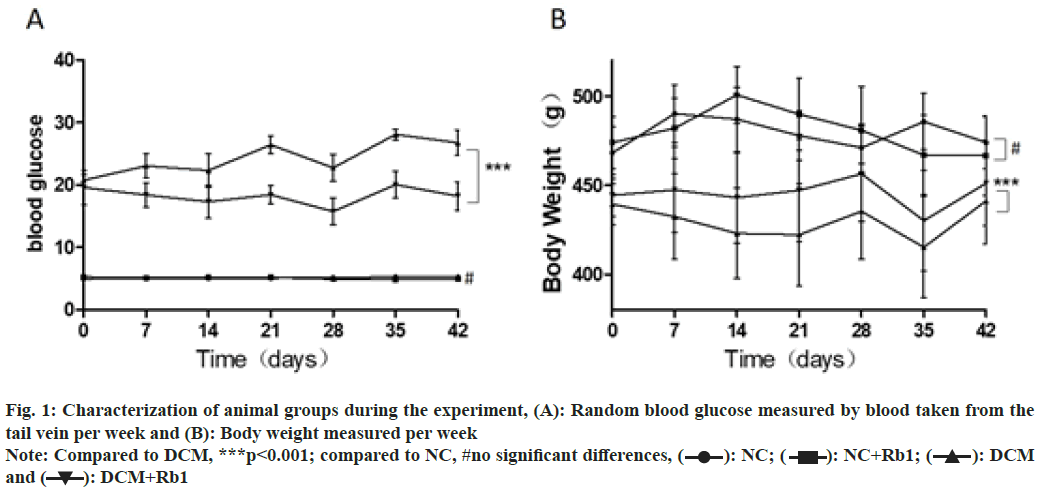
When the DCM model was induced, the rats in both diabetic groups displayed severe hyperglycemia, lower body weight and higher levels of total cholesterol and triglycerides compared to non-diabetic groups as shown in Table 1. We also measured body weight and blood glucose weekly during the study. Comparing the diabetic group treated with ginsenoside Rb1 to the group treated only with insulin, we found that ginsenoside Rb1 significantly suppressed hyperglycemia and weight loss (p<0.001). Meanwhile, ginsenoside Rb1 had no obvious effects on normal rats measured by blood glucose and body weight as shown in fig. 1.
| Blood glucose (mmol/l) | Body weight (g) | Total cholesterol (mmol/l) | Triglyceride (mmol/l) | |
|---|---|---|---|---|
| DCM | 20.3±2.5*** | 444.3±36.5*** | 16.3±3.9*** | 6.3±1.9*** |
| NC | 6.2±1.3 | 485.8±22.5 | 4.6±0.5 | 0.9±0.3 |
Note: Compared to NC, ***p<0.001
Table 1: Characterization of Animal Groups
At the end of the experiment, the rats in each group underwent cardiac ultrasound to evaluate cardiac performance. The results showed that the LVEDD and LVESD of the heart tissue in the DCM group were smaller than in the control group. Meanwhile, the myocardial echo in the DCM group was uneven and disordered. Thus, ginsenoside Rb1 improved the cardiac function of DCM rats but had no obvious effects on the heart tissue of normal rats as shown in fig. 2 and Table 2.
| LVEDD (mm) | LVEDD index | LVESD (mm) | LVESD index | LVEF (%) | FS (%) | |
|---|---|---|---|---|---|---|
| NC | 6.3±0.7 | 14.6±1.4 | 3.9±0.4 | 8.5±0.5 | 79.8±3.4 | 51.3±3.0 |
| Rb1 | 6.4±0.8 | 14.8±1.0 | 3.8±0.3 | 7.9±0.6 | 80.4±4.9 | 50.3±3.7 |
| DCM | 5.5±0.5 | 21.3±2.1*** | 4.0±0.5 | 14.0±1.7*** | 67.6±6.0*** | 38.8±2.7*** |
| DCM+Rb1 | 6.2±0.5 | 19.8±1.5 | 3.9±0.4 | 10.4±1.4*** | 71.3±6.3### | 44.6±3.8### |
Note: Compared to NC, ***p<0.001 and compared to DCM, ###p<0.001
Table 2: Cardiac Performance by Cardiac Ultrasound
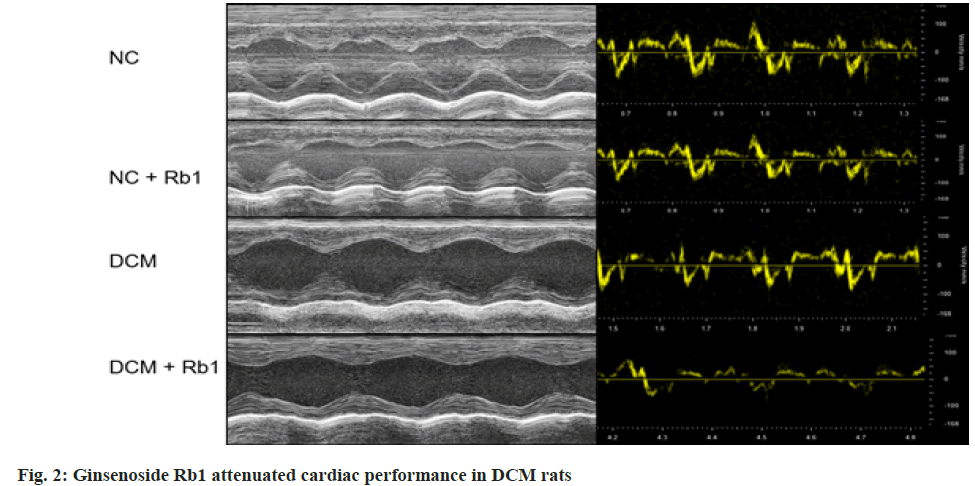
At the end of the experiment, cardiac fibrosis was measured by HE and Masson staining in the myocardial tissue. The myocardial cells in the control group were neatly arranged with uniform nuclei and tightly connected and the myocardial cells in the DCM group were swollen, disordered and fibrotic. Ginsenoside Rb1 reduced the pathological changes of myocardial tissue in DCM rats, but had no significant effect on the myocardial tissue of normal rats as shown in fig. 3.
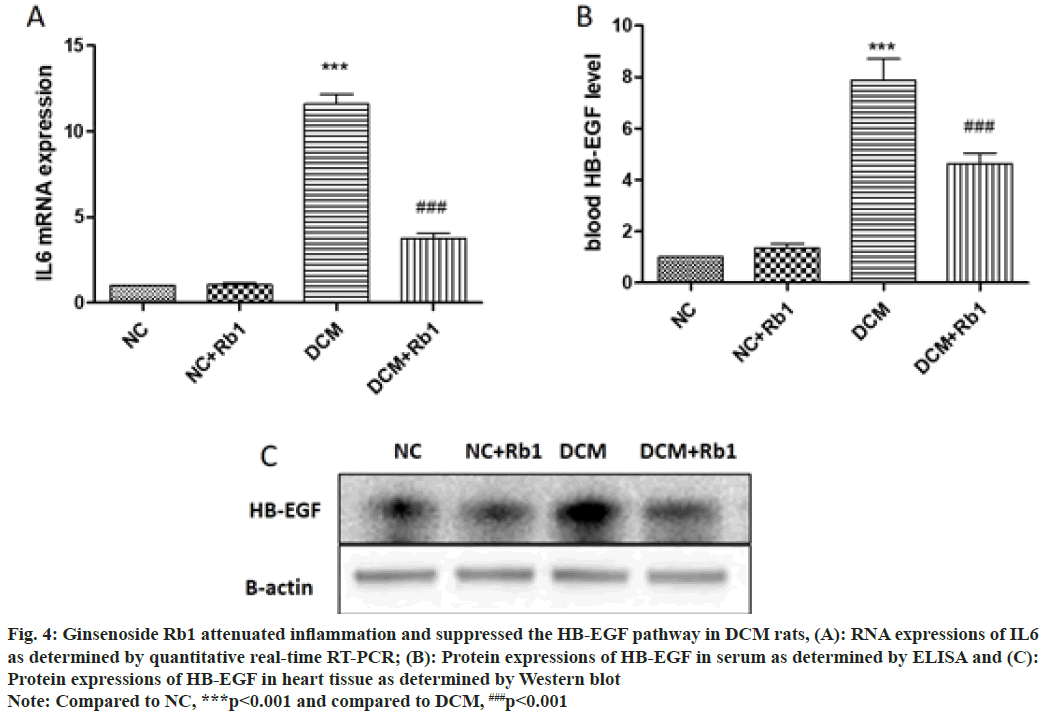
At the end of the experiment, cardiac inflammation was measured by the expression of IL-6 messenger RNA (mRNA) in the myocardial tissue. IL-6 levels in the DCM group were significantly higher than in the control group, but ginsenoside Rb1 suppressed IL-6 expression in DCM rats. Meanwhile, it had no significant effect on IL-6 expression in normal rats as shown in fig. 4A.
Fig. 4: Ginsenoside Rb1 attenuated inflammation and suppressed the HB-EGF pathway in DCM rats, (A): RNA expressions of IL6
as determined by quantitative real-time RT-PCR; (B): Protein expressions of HB-EGF in serum as determined by ELISA and (C):
Protein expressions of HB-EGF in heart tissue as determined by Western blot
Note: Compared to NC, ***p<0.001 and compared to DCM, ###p<0.001
At the end of the experiment, the blood samples from each group of rats were examined for HB-EGF by ELISA. The results were serum HB-EGF level of rats in the DCM group was significantly higher than that of the control group; ginsenoside Rb1 appeared to suppress serum HB-EGF levels in DCM rats. In addition, it had no significant effect on serum HB-EGF levels in normal rats as shown in fig. 4B. The expression of HB-EGF in the myocardial tissue was measured by Western blot, with similar results to the serum HB-EGF as shown in fig. 4C.
At the end of the experiment, the serum Creatine Kinase-MB (CK-MB), Alanine Transaminase (ALT) and Creatinine (Cr) levels of DCM rats were significantly higher than those of the control group. However, ginsenoside Rb1 apparently reduced these levels in diabetic rats. Ginsenoside Rb1 had no obvious effect on the serum CK-MB, ALT and Cr levels of normal rats. The results also indicated that ginsenoside Rb1 had no significant liver, kidney or cardiotoxicity at the selected dose as shown in Table 3.
| ALT (U/l) | Cr (U/l) | CK-MB (U/l) | |
|---|---|---|---|
| NC | 31.9±5.9 | 50.8±6.9 | 540.2±53.2 |
| Rb1 | 29.6±6.4 | 50.0±7.3 | 562.7±59.4 |
| DCM | 114.6±36.7*** | 54.4±8.9 | 754.7±115.6*** |
| DCM+Rb1 | 71.4±16.6### | 54.7±11.8 | 625.7±108.9### |
Note: Compared to NC, ***p<0.001 and compared to DCM, ###p<0.001
Table 3: Safety of Ginsenoside Rb1 Treatment
DCM is one of the most serious cardiovascular complications of diabetes and is highly correlated with the incidence of heart failure and patient mortality. It is difficult to prevent DCM because of unclear pathogenesis. However, diabetes is considered an inflammatory disease and inflammatory factors play an important role in the development of diabetes and its cardiovascular complications[14-18]. In the present study, the inflammatory signaling pathway in the myocardial tissue of the DCM group was significantly activated, showing that the level of inflammatory factor (IL6) mRNA was significantly increased. Meanwhile, the ginsenoside Rb1 intervention can relieve the inflammatory response in DCM rats, suggesting that ginsenoside Rb1 may have the effect of inhibiting the inflammatory response while simultaneously helping insulin to lower blood glucose. Myocardial fibrosis is another characteristic pathological change in DCM. In the diabetic state, a large amount of collagen deposition in the interstitium of the myocardium increases the stiffness of the ventricle and decreases the compliance, which may eventually lead to ventricular diastolic and systolic dysfunction[19-22]. In this study, we also observed that the degree of myocardial fibrosis in diabetic rats treated with ginsenoside Rb1 was reduced.
HB-EGF is named for its role in promoting growth and is abnormally activated in diabetes and atherosclerosis[23]. It has been reported that high blood glucose can lead to elevated HB-EGF and thus to abnormal phosphorylation of HB-EGFR, resulting in oxidative stress and endothelial dysfunction[24]. STZ induced mouse diabetes models with subsequent endothelial dysfunction have been widely reported[25-27]. In addition, the serum HB-EGF concentration of diabetic patients is higher than that of normal patients, which is consistent with previous studies[28-30]. Our results confirm that the abnormal expression of HBEGF in the diabetes model plays an important role in cardiovascular complications. Meanwhile, inhibition of HB-EGFR activation[25,31,32] or reduction of insulin resistance[33] can reverse these phenomena. Our results indicate that HB-EGF levels in the myocardium and serum of DCM rats are significantly increased and that ginsenoside Rb1 can reduce them effectively.
Rb1 ginsenosides are extracts of the traditional Chinese medicinal herb ginseng and more information is needed on their safety. We measured serum CK-MB levels to assess heart injury, serum Cr levels to assess kidney injury and serum ALT levels to assess liver injury. The results reveal that the levels of serum CK-MB, Cr and ALT in the DCM group were significantly higher than those in the Normal Control (NC) group, while ginsenoside Rb1 intervention reduced serum CKMB, Cr and ALT levels. Meanwhile, in the NC group, ginsenoside Rb1 intervention had no significant effect on serum CK-MB, Cr or ALT levels. These results indicate that ginsenoside Rb1 has no obvious toxicity or side effects in a rat's heart, liver or kidneys at the selected dose in this research and is a safe drug of choice.
In summary, ginsenoside Rb1 treatment significantly attenuates cardiac inflammation and fibrosis by inhibiting the activity and expression of HB-EGF, leading to improved cardiac function in DCM rats. Our results indicate that ginsenoside Rb1 might have therapeutic potential for the prevention and treatment of DCM.
Acknowledgements:
This research was supported by Jinhua Science and Technology Project (No: 2018-4-051).
Ethical approval and consent to participate:
All methods were carried out in accordance with relevant guidelines and regulations. And this article was approved by the Ethics Committee of Jinhua people’s Hospital.
Conflict of interests:
The authors declared no conflicts of interest.
References
- Thurmond DC, Gaisano HY. Recent insights into beta-cell exocytosis in Type 2 diabetes. J Mol Biol 2020;432(5):1310-25.
[Crossref] [Google Scholar] [Pub Med]
- Yang W, Lu J, Weng J, Jia W, Ji L, Xiao J, et al. Prevalence of diabetes among men and women in China. N Engl J Med 2010;362(12):1090-101.
[Crossref] [Google Scholar] [Pub Med]
- Mauricio D, Alonso N, Gratacòs M. Chronic diabetes complications: The need to move beyond classical concepts. Trends Endocrinol Metab 2020;31(4):287-95.
[Crossref] [Google Scholar] [Pub Med]
- Members ATF, Rydén L, Grant PJ, Anker SD, Berne C, Cosentino F, et al. ESC guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD: The task force on diabetes, pre-diabetes, and cardiovascular diseases of the European Society of Cardiology (ESC) and developed in collaboration with the European Association for the Study of Diabetes (EASD). Eur Heart J 2013;34(39):3035-87.
[Crossref] [Google Scholar] [Pub Med]
- Alonso N, Moliner P, Mauricio D. Pathogenesis, clinical features and treatment of diabetic cardiomyopathy. Heart Fail Res Clin Pract 2017;1067:197-217.
[Crossref] [Google Scholar] [Pub Med]
- Kim JH. Pharmacological and medical applications of Panax ginseng and ginsenosides: A review for use in cardiovascular diseases. J Ginseng Res 2018;42(3):264-9.
[Crossref] [Google Scholar] [Pub Med]
- Xiong Y, Shen L, Liu KJ, Tso P, Xiong Y, Wang G, et al. Antiobesity and antihyperglycemic effects of ginsenoside Rb1 in rats. Diabetes 2010;59(10):2505-12.
[Crossref] [Google Scholar] [Pub Med]
- Waki I, Kyo H, Yasuda M, Kimura M. Effects of a hypoglycemic component of ginseng radix on insulin biosynthesis in normal and diabetic animals. J Pharmacobiodyn 1982;5(8):547-54.
[Crossref] [Google Scholar] [Pub Med]
- Zhong ZD, Wang CM, Wang W, Shen L, Chen ZH. Major hypoglycemic ingredients of Panax notoginseng saponins for treating diabetes. Sichuan Da Xue Xue Bao Yi Xue Ban 2014;45(2):235-9.
- Shang W, Yang Y, Jiang B, Jin H, Zhou L, Liu S, et al. Ginsenoside Rb1 promotes adipogenesis in 3T3-L1 cells by enhancing PPARγ2 and C/EBPα gene expression. Life Sci 2007;80(7):618-25.
[Crossref] [Google Scholar] [Pub Med]
- Xie Z, Truong TL, Zhang P, Xu F, Xu X, Li P. Dan-Qi prescription ameliorates insulin resistance through overall corrective regulation of glucose and fat metabolism. J Ethnopharmacol 2015;172:70-9.
[Crossref] [Google Scholar] [Pub Med]
- Yu X, Ye L, Zhang H, Zhao J, Wang G, Guo C, et al. Ginsenoside Rb1 ameliorates liver fat accumulation by up regulating perilipin expression in adipose tissue of db/db obese mice. J Ginseng Res 2015;39(3):199-205.
[Crossref] [Google Scholar] [Pub Med]
- Shang W, Yang Y, Zhou L, Jiang B, Jin H, Chen M. Ginsenoside Rb1 stimulates glucose uptake through insulin-like signaling pathway in 3T3-L1 adipocytes. J Endocrinol 2008;198(3):561-9.
[Crossref] [Google Scholar] [Pub Med]
- Stratmann B, Tschoepe D. The diabetic heart: Sweet, fatty and stressed. Expert Rev Cardiovasc Ther 2011;9(9):1093-6.
- Navab M, Gharavi N, Watson AD. Inflammation and metabolic disorders. Curr Opin Clin Nutr Meta Care 2008;11(4):459-64.
- Wellen KE, Hotamisligil GS. Inflammation, stress and diabetes. J Clin Invest 2005;115(5):1111-9.
[Crossref] [Google Scholar] [Pub Med]
- Dandona P, Aljada A, Bandyopadhyay A. Inflammation: The link between insulin resistance, obesity and diabetes. Trends Immunol 2004;25(1):4-7.
[Crossref] [Google Scholar] [Pub Med]
- Donath MY, Shoelson SE. Type 2 diabetes as an inflammatory disease. Nat Rev Immunol 2011;11(2):98-107.
[Crossref] [Google Scholar] [Pub Med]
- Yang B, Li M, Shi ZG, Feng QZ. Bosentan ameliorates the expression of fibrotic related growth factors and collagen-1 in diabetic mice. Anadolu Kardiyol Derg 2012;12(8):621-7.
[Crossref] [Google Scholar] [Pub Med]
- Teupe C, Rosak C. Diabetic cardiomyopathy and diastolic heart failure-difficulties with relaxation. Diabetes Res Clin Pract 2012;97(2):185-94.
[Crossref] [Google Scholar] [Pub Med]
- Boudina S, Abel ED. Diabetic cardiomyopathy revisited. Circulation 2007;115(25):3213-23.
[Crossref] [Google Scholar] [Pub Med]
- Zhang Y, Edgley AJ, Cox AJ, Powell AK, Wang B, Kompa AR, et al. FT011, a new anti-fibrotic drug, attenuates fibrosis and chronic heart failure in experimental diabetic cardiomyopathy. Eur J Heart Fail 2012;14(5):549-62.
[Crossref] [Google Scholar] [Pub Med]
- Mindur JE, Swirski FK. Growth factors as immunotherapeutic targets in cardiovascular disease. Arterioscler Thromb Vasc Biol 2019;39(7):1275-87.
- Gilbert RE, Rumble JR, Cao Z, Cox AJ, van Eeden P, Allen TJ, et al. Endothelin receptor antagonism ameliorates mast cell infiltration, vascular hypertrophy and epidermal growth factor expression in experimental diabetes. Circ Res 2000;86(2):158-65.
[Crossref] [Google Scholar] [Pub Med]
- Akhtar S, Yousif MH, Dhaunsi GS, Sarkhouh F, Chandrasekhar B, Attur S, et al. Activation of ErbB2 and downstream signalling via Rho kinases and ERK1/2 contributes to diabetes-induced vascular dysfunction. PLoS One 2013;8(6):e67813.
[Crossref] [Google Scholar] [Pub Med]
- Elms S, Toque H, Rojas M, Xu Z, Caldwell R, Caldwell RB. The role of arginase I in diabetes-induced retinal vascular dysfunction in mouse and rat models of diabetes. Diabetologia 2013;56(3):654-62.
[Crossref] [Google Scholar] [Pub Med]
- Ng HH, Leo CH, Prakoso D, Qin C, Ritchie RH, Parry LJ. Serelaxin treatment reverses vascular dysfunction and left ventricular hypertrophy in a mouse model of type 1 diabetes. Sci Rep 2017;7(1):39604.
[Crossref] [Google Scholar] [Pub Med]
- Sundar NM, Krishnan V, Krishnaraj S, Hemalatha V, Alam MN. Comparison of the salivary and the serum nitric oxide levels in chronic and aggressive periodontitis: A biochemical study. J Clin Diagn Res 2013;7(6):1223-7.
[Crossref] [Google Scholar] [Pub Med]
- Mirhafez SR, Pasdar A, Avan A, Esmaily H, Moezzi A, Mohebati M, et al. Cytokine and growth factor profiling in patients with the metabolic syndrome. Br J Nut 2015;113(12):1911-9.
[Crossref] [Google Scholar] [Pub Med]
- Loukovaara M, Leinonen P, Teramo K, Andersson S, Alfthan H, Stenman UH. Diabetic pregnancy associated with increased epidermal growth factor in cord serum at term. Obstetrics Gynecol 2004;103(2):240-4.
[Crossref] [Google Scholar] [Pub Med]
- Benter IF, Benboubetra M, Hollins AJ, Yousif MH, Canatan H, Akhtar S. Early inhibition of EGFR signaling prevents diabetes-induced up-regulation of multiple gene pathways in the mesenteric vasculature. Vascul Pharmacol 2009;51(4):236-45.
[Crossref] [Google Scholar] [Pub Med]
- Palen DI, Matrougui K. Role of elevated EGFR phosphorylation in the induction of structural remodelling and altered mechanical properties of resistance artery from type 2 diabetic mice. Diabetes Meta Res Rev 2008;24(8):651-6.
[Crossref] [Google Scholar] [Pub Med]
- Fountas A, Diamantopoulos LN, Tsatsoulis A. Tyrosine kinase inhibitors and diabetes: A novel treatment paradigm? Trends Endocrinol Metab 2015;26(11):643-56.
[Crossref] [Google Scholar] [Pub Med]