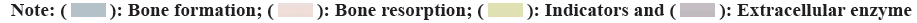- *Corresponding Author:
- Xiaming Cai
Department of Endocrinology, Affiliated Hospital of Putian University, Putian, Fujian Province 351100, China
E-mail: 13950707296@139.com
| Date of Received | 05 January 2023 |
| Date of Revision | 10 July 2023 |
| Date of Acceptance | 02 December 2023 |
| Indian J Pharm Sci 2023;85(6):1594-1604 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
Osteoporosis has gradually become a common human disease. The main cause of this common disease is caused by the degradation of the internal structure of bone and soft tissues, which causes great troubles to the lives of elderly patients. Quercetin is a flavonoid drug that is commonly used to treat osteoporosis. Therefore, it is very important to study the pharmacological and clinical effects of quercetin. The purpose of this article is to solve some problems in the clinical application of quercetin and to explore the effect of quercetin on bone metabolism and serum osteocalcin in osteoporotic rats. Taking 90 rats as the research object, an animal model of osteoporosis was constructed. The rats were randomly divided into sham operation control group, ovariectomy control group and quercetin observation group. Except for the sham operation group, all rats were subjected to ovariectomy, and the rats in the quercetin observation group were given quercetin drug intervention. 1 w later, all rats were tested for bone metabolism and serum osteocalcin levels, and related data were recorded and analyzed. The results of the study showed that with the intervention of quercetin, the bone metabolism index of osteoporosis rats increased from the original (53.49±3.41) to (86.27.2±4.22), and the serum calcium level from the original (16.28±0.56) μg/l increased to (37.64±2.35) μg/l, and the bone mineral density and bone tissue structure of rats also improved accordingly. It can be seen that quercetin can promote bone metabolism in osteoporotic rats, increase the content of osteocalcin in rats, and have a good effect on the prevention and treatment of osteoporosis.
Keywords
Quercetin, osteoporosis, bone metabolism, serum osteocalcin
Osteoporosis is a systemic bone injury disease. The amount of spine bones per unit body volume is greatly reduced, and the microstructure of skeletal soft tissue is absolutely degraded. The risk of occurrence has greatly increased. The main early clinical manifestations of early osteoporosis patients are pain, spinal joint deformity, and easy fracture. Severe pain will greatly reduce the quality of daily life of epilepsy patients. Excessive deformation of the spine may severely limit the normal activity of the spine patient. Osteoporosis can cause lifelong disability for many patients, which not only seriously affect the quality of daily life of patients, but also bring a serious social and economic burden to our society, families and other personal lives. The current clinical research on the etiology and mechanism of chronic osteoporosis patients mainly focuses on the abnormal metabolism of calcium in the body. Abnormally insufficient calcium intake can easily lead to chronic osteoporosis, that is, low calcium causes osteoporosis. Clinical treatment with calcium chloride, active phosphorus and vitamins C and D, sex hormones, calcitonin, diphosphate and dihydrouridine, although it has a certain clinical effect, but the cost of medication is expensive and the side effects are large. The latest medical research results have found that quercetin has a certain medical prevention and health care effect on elderly osteoporosis patients.
Quercetin is a natural plant alkaloid estrogen with strong estrogen antagonist activity[1]. As we all know, quercetin is widely distributed in the plant kingdom and has the biological activities of a variety of enzymes, which is increasingly widely valued by modern people. In recent years, researchers have successively discovered that peanut quercetin has many new biological activities, including its antibacterial, anti-malignant, anti-depressant, anti-diabetic, hepatoprotective, and atherosclerotic effects[2]. In addition, quercetins also have the activity of inhibiting and increasing the induction of osteoblast white matter differentiation, which can effectively inhibit the increase of osteoclast bone white matter absorption, increase osteoblast differentiation-mediated bone cell formation and induce osteoclast apoptosis. Between mediated bone cell resorption, the important basis of the cellular homeostasis and structural stability of bone metabolism is also the most important factor that may lead to the occurrence of chronic osteoporosis diseases[3]. Therefore, it is of great significance to explore the effect mechanism of quercetin on bone metabolism and serum osteocalcin.
In order to explore the effect of quercetin on bone metabolism and serum osteocalcin in osteoporotic rats, a large number of related materials were consulted. Among them, Jo et al.[4] gave a detailed introduction to the pharmacological effects of quercetin, discussed the scope of clinical application of quercetin, and some side effects brought about by quercetin, emphasizing the precautions for using quercetin. Poshke elaborated the quercetin drug composition and the extraction method of quercetin, which improved the traditional extraction technology and promoted the industrial production of quercetin[5]. Qu et al.[6] pointed out that in the case of today’s medical technology is very limited, drug combination therapy is the main treatment for osteoporosis, while emphasizing that quercetin as a common drug for the treatment of osteoporosis will have certain side effects, and cannot be cured, this state quo is the bottleneck of osteoporosis treatment. Wu et al.[7] elaborated the etiology and pathology of the osteoporosis disease and discussed the current academic issues about osteoporosis disease, analyzed the current research status and research significance, and provided a theoretical basis for the treatment of osteoporosis. Jeong et al.[8] learned from related experiments that the flavonoid quercetin can regulate the bone structure of the human body, increase bone tissue density, and can improve bone metabolism and serum osteocalcin levels, which has a certain effect on the treatment of osteoporosis.
In the study of the effect of quercetin on bone metabolism and serum osteocalcin in osteoporotic rats, this article summarizes and analyzes the research experience and achievements of a large number of predecessors. In addition, this article has made some innovations in the research content and research methods. The specific innovations are as follows; first, this article is the first observation and detection of bone tissue density, bone structure, bone metabolism and serum osteocalcin in rats using transmission electron microscopy and Polymerase Chain Reaction (PCR) fluorescence measurement, which improves the efficiency of observation and detection. Second, this article is the first to use the multi-component statistical data analysis method collected by data analysis software, and the statistical data collection and analysis of observation results using Statistical Package for the Social Sciences (SPSS) 26.0 software, which greatly improves the accuracy of the research results. Third, this article is the first to analyze the effect of quercetin on osteoporosis in rats through the ovariectomized low calcium diet Sprague–Dawley (SD) rat osteoporotic fracture model, from the changes of bone metabolism and serum osteocalcin in rats.
Materials and Methods
Etiology and pathology of osteoporosis:
The main clinical pathological structural changes of osteoporosis are mainly the significant reduction of the bone matrix and the content of mineral elements in the bone. In the early stage of osteoporosis, the anatomical observation of the transverse and longitudinal sections of the human body growing bone and soft tissue and the vertebral body. The anatomical observation of the pelvis clearly showed changes in the structure of the cartilage cortex[9]. This is mainly caused by the osteoclast’s gradual differentiation and absorption of surface fat inside the bone soft cortex. Under normal circumstances, the activation and transformation of osteoblasts is normal, but the active transformation and activation of osteoclasts is abnormal, which increases the number of osteoclasts and increases bone resorption. At the same time, the volume of cancellous trabecular bone smaller and thinner, the number is reduced by up to 40 %. Bone movement must have sufficient movement stiffness and certain toughness to maintain the strength of the bone so that it can withstand external forces and avoid fractures. Therefore, all bone types need to be complete. The spiral layer includes a 3-chain longitudinal spiral layer rich in collagen, non-collagen and a large amount of dodecyl hydroxyapatite. By repeating the time, the repeated processes of spatiotemporal bone resorption and bone formation can maintain the integrity of the bone structure. This iterative process is also known as "bone reconstruction". Bone reconstruction in adulthood can balance and maintain a certain number of bones. As bone age continues to increase, bone formation, bone mass and resorption are negatively balanced, and bone imbalance will cause bone loss during reconstruction[10].
Human bone tissue cells can stimulate physiological changes or microbial destruction through bone lamellar bone or microvascular nervous system, of which osteoclasts account for about 10 %-20 % of human bone resorption cells. They are formed by the cleavage, differentiation and binding of the front-end receptors of single phagocytes responsible for bone resorption. Osteoclast differentiation can effectively activate Nuclear Factor-Kappa B (NF-κB) and promote the normal differentiation of precursor osteoclasts. The normal proliferation and survival function of osteoclasts depends on the interaction of Macrophage-Colony Stimulating Factor (M-CSF) and c-FMS. The bone marrow protective hormone phosphate (Osteoprotegerin (OPG)) secreted by osteoblasts is also an activity-inhibiting receptor that can be used as the Receptor Activator of NF-kB Ligand (RANKL) (a visible fat-soluble substance in the body) and can compete with various other competitive osteoclast receptors. They combine to form the RANKL receptor, which effectively inhibits the activity of other osteoclasts in the body. After bone resorption cells are important precursors for the rapid differentiation of these osteoblasts, these osteoblasts can be quickly recruited by simultaneously detecting the normal rate of change of the transforming growth factor gradient of bone cell Transforming Growth Factor-Beta 1 (TGF-β1). Osteoblasts are composed of mesenchymal filled osteoblastic stem cells, pituitary osteoblasts and nuclei, which mainly form bone matrix. With the continued mineralization of bone matrix, osteoblasts can also develop into osteoblasts, which can be wrapped or buried in the basic layer of bone matrix or left on the surface of bone tissue. The calculation method for the number of osteoblasts in human bone tissue is shown in equation 1.
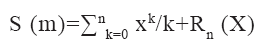
Where S (m) represents the number of osteoblasts and R and X represent the expression coefficient of bone matrix.
The main reason for osteoporosis is that the immunosuppressive activity of estrogen on osteoclast tissue cells is significantly reduced, and the number of osteoclast apoptosis is significantly reduced. As the number of cells increases, the number of apoptotic cells decreases, life span increases, and bone resorption and contraction greatly increase. The strength of osteoblasts on both sides and the formation of bone and joint formation also increased significantly, but it is not necessarily sufficient to effectively compensate for excessive bone resorption on both sides[11]. Active remodeling of bones and joints, and unbalanced movements can also cause cancellous bone on both sides, with joints thinning or severely broken. Lower levels of estrogen may reduce the physiological sensitivity of human bones to dynamic changes. Therefore, the human bone structure has similar pathological features as the loss of radioactive bone hormone excreted by the human body. The severe occurrence of osteoporosis and related fractures is usually an inevitable result of the interaction of natural and unnatural genetic pathogens. Genetic factors mainly include human bone size, bone mass, structure, microstructure and internal tissue characteristics.
Pharmacological effects of quercetin in bone diseases:
Quercetin is an organic compound containing flavonoid sterols. It is widely believed to exist in many traditional Chinese medicines such as beef tendon, licorice, safflower, mulberry parasite, and epicedium. Quercetin at different drug concentrations has different promoting effects on the benign proliferation of rabbit cartilage epithelial cells induced by Interleukin-1β (IL-1β) kinase, and their promoting effects are directly proportional to different concentrations. The effects of Extracellular Regulated Kinase (ERK) signaling pathway on bone formation of bone Marrow Mesenchymal Stem Cells (MSCc) in SD rats were observed through experiments. The results show that a certain concentration of quercetin can promote the proliferation of MSCs. Osteogenic differentiation and ERK signal activation play a very important role in the osteogenesis of MSCs[12]. Quercetin can induce apoptosis of MG-68 cells through the caspase-3 pathway, and is dose-dependent and time-dependent, suggesting that quercetin’s anti-tumor mechanism may interact with caspase-3 to induce apoptosis of MG-68 cells death. Quercetin at different concentrations has an inhibitory effect on the proliferation of human chondrosarcoma cells (SW5) and has significant concentration dependence.
The active expression inhibits the absorption of bone calcium and prevents chronic osteoporosis. Through clinical studies on bone cells in vitro, we found that quercetin can inhibit the differentiation of mature osteoclasts and progenitor cells toward the direction of pocks, and can disrupt the circulation of myofibrils of mature osteoclasts and inhibit the absorption of bone marrow. Quercetin drugs can significantly promote the growth and proliferation of human osteoblasts and bone mineralization, thereby effectively enhancing the bone formation activity of human osteoblasts. Rutan can promote the proliferation of Rat Osteoblasts (ROB) cells, but it has no effect on mineralization, but it can inhibit Alkaline Phosphatases (ALPs) secretion activity, and has basically no promotion effect on the formation of ROB. The glycoside produced by 3-OH in quercetin reduces the activity of osteoblasts cultured in vitro. It is speculated that glycoside may play an anti-osteoporosis role through its metabolite quercetin, which stimulates the formation of osteoblast flavonoids. And realization, as the active ingredient of commonly used Chinese herbal medicine in orthopedics, shows certain advantages in regulating bone metabolism. Based on the above pharmacological effects, the medicinal properties of quercetin have a protective effect on bone loss after menopause. As an estrogen receptor modulator, it can bind to estrogen receptor-related parts and regulate many substrates and signaling pathways downstream of the estrogen receptor, but it plays a different role in the antagonistic or agonistic effects of different receptors.
Quercetin can also be involved in inhibiting the activation of cell genes that regulate cell regeneration and enhance nutritional metabolism support, increase the gene expression of upstream ribonucleotide adenosine and gene activation of downstream neural channels, and activate regenerative motor nerves on peripheral axons. Increase the speed of motor regenerative neurons and transducers in the foot and the local muscle regulation function of the plantar. Quercetin decoction can significantly reduce the inflammation of the tibial joint mucosa and the swelling between the tibial joints in rats with acute and gouty tibial arthritis. In typical mice with gouty induced by sodium urate dorsal (Monosodium Urate (MSU)) crystals, quercetin can inhibit the local mechanical mild pain induced by MSU crystals as well as allergies, positive aggregation of white blood cells, Tumour Necrosis Factor Alpha (TNF-α) and IL-1β. The production of IL-1β, super deoxy negative cations, etc., reduces the body’s antioxidant effect level, the expression of NF-κB inhibits the formation of microRNA (miRNA) in the inflammatory response complex. Quercetin effectively reduced the ratio of (I) an MMP13 inhibitor (iMMP13)/ Tissue Inhibitors of Metalloproteinases (TIMPs), promoted and inhibited the degradation of amino acid outside the bone and cartilage joint cells, and protected the bone and cartilage. Quercetin cannot only significantly inhibit the bone white matter metabolism of early osteoporosis and rats, promote the formation of bones, but also have a significant adverse effect on the dynamics of bone microbes.
Related experiments of quercetin on rats:
Object selection and grouping: In this experiment, 90 rats with a 5 mo old body weight close to each other were selected as the research object, and the body weight was (270±5.0) g. The sterilized and non-polluting laboratory was selected as the experimental site. The laboratory temperature was 24°-29° the moisture in the air is about 18.3 %-22.8 %, the exposure time of the sun is 7:00-18:00, the day lighting rate is good, the laboratory is kept ventilated to ensure that the oxygen content in the air is sufficient, according to the national standard whistle animal feeding specifications. For cage feeding, five rats were randomly placed in the same cage to allow rats to eat and drink freely. The feeding period was 1 w. Before the experiment, all rats were checked for health, and unhealthy rats were excluded.
Related materials:
The main equipments used in this experimental research institute were; bone density measuring instrument, automatic biochemical instrument, electronic biomechanical measuring instrument, YSK-2009ER transmission electron microscope (Japan). PCR fluorescence amplification instrument (Jiangsu Haupia Medical Instrument Factory), pathology slicer (Jiangsu Zhongdu Analytical Instrument Co, Ltd.), 146HS photometric measuring instrument (Jiangsu Zhongdu Analytical Instrument Co, Ltd.), HGP constant temperature water bath (Shanghai Pudong Scientific Instrument Co, Ltd.). BCL-210A Rongcheng water tank (Jiangsu Keitai Electric Co., Ltd.). Vertical electrophoresis instrument, electronic balance produced by Stayer Company, room temperature high-speed centrifuge.
The main reagents used in the experiment are as follows; quercetin 200 g, rat serum Bromocresol Green (BCG) reagent 300 ml, rat monoclonal antibody, ophthalmology immune antibody, Salmonid Alphavirus (SAV) immunohistochemistry kit. Color reagent 500 ml, collagen 120 mg (purchased from NET company), protease inhibitor (laboratory stock), protein phosphatase inhibitor 80 ml, paraformaldehyde 20 ml, ammonium bicarbonate 40 ml. Permanent Magnet Synchronous Generator (PMSG) (purchased from Yunnan Anuran Chemical Co, Ltd.), osteocalcin 200 ml. Poly Lactide-Glycolide (PLG) biological tissue solvent (Shanghai Chemical Reagent Factory). Other auxiliary equipment and reagents are shown in Table 1.
| Group | Usage amount | Source |
|---|---|---|
| Fluorescence amplifier | 1 | Xiyu Technology Company |
| High-speed centrifuge | 1 | Foreign Technology Company |
| Paraformaldehyde | 200 ml | Shanghai Analytical Instruments |
| Collagen | 500 ml | Jiangsu Feng Hua |
| Hydrochloric acid | 1 | Gaohu Chemical Enterprise |
| Sulfuric acid | 300 mg | Japan Sanwa Kimono |
| Sodium chloride | 750 ml | American SGH |
| Electrophoresis | 1 | Bosch, Germany |
Table 1: Other Auxiliary Equipment and Reagents
Animal model of osteoporosis in rats:
After the grouping was completed, 30 % chloral hydrate anesthesia was used for abdominal anesthesia and skin preparation was disinfected. A longitudinal incision of approximately 1 cm was taken from the 5th lumbar spine at the midline of the abdomen and separated into the abdominal cavity in sequence, looking for a pink bilateral ovary along the inner wall of the bilateral fallopian tubes. Elastic tissue, strict surgical ligation of bilateral fallopian tube tissue and its surrounding capillaries, fatty elastic tissue, complete surgical removal of bilateral elastic ovaries, sham surgery ovarian tissue can remove an equal amount of vascular fatty elastic tissue around the bilateral ovaries, no surgery. Bilateral ovaries were removed, the abdominal cavity of the uterus was closed, and sutures were made layer by layer or without interruption. After the operation, cephalosporin 3000 µg/kg could be injected intramuscularly to prevent ovarian infection. The rats were anesthetized by intraperitoneal injection of 0.5 g/kg with ketamine hydrochloride injection, and the drug in the prone position was fixed on the operating table of the rat. Afterwards, a circular incision about 1 cm in length is made in the back of the mouse thigh near the hip of the rat, and the separation is performed layer by layer. The sciatic nerve can be found between the semi-key muscle, semimembranosus muscle, and biceps femoris on both sides. Close to the upper segment, excise 2.0 cm, then suture the skin incision, sciatic nerve is isolated and sutured, the osteoporosis model is completed.
Quercetin drug intervention methods:
1 w after the establishment of the rat osteoporosis animal model, all rats were intragastrically administered with normal saline twice a day, 5 ml each time. The ovariectomized control group underwent ovarian removal surgery. The rats in the quercetin observation group were given medical intervention, and the rats in the observation group were garaged at an average of 150 mg/kg quercetin reagent three times a week. The drug intervention lasted a total of 2 mo.
Determination of serum osteocalcin levels:
After drug intervention, intraperitoneal injection of 2 % pentobarbital sodium solution to anesthetized rats, use a small scalpel or scissors to quickly cut the abdominal cavity of the rat, and use an intravenous syringe to quickly remove 5 ml of venous blood in the cavity below room temperature place for 30 min, centrifuge at 4000 rpm for 20 min at 10° (centrifugal outlet radius 20 cm), take rat serum at -30° and store it in the refrigerator. This method simultaneously detects osteocalcin sodium in the serum of rats in each group. After taking blood, a large number of mice with bleeding in the abdomen or aorta were executed. The bilateral maxillary femur, the right maxillary femur, the proximal end bone and the metaphysis bone were implanted and methyl methacrylate was implanted without calcium loss. Paraffin section, 12 μm thick, Hematoxylin and Eosin (HE) stained slightly under laser microscope to observe the abnormal shape of bone soft tissue. Take the proximal metaphyseal bone tissue of the left femur and determine the level of serum osteocalcin in rat bone tissue by PCR fluorescence detection.
Detection of bone metabolism in rats:
All rats were anesthetized after receiving drug injection intervention for 2 mo. Take the laparoscopic aortic blood, centrifuge at room temperature for 30 min, 5000 rpm, centrifuge at a radius of 10 cm, for 18 min to separate the test serum, and use the combined Enzyme-Linked Immunosorbent Assay (ELISA) kit and vascular microplate cell reading filler to determine. Osteocalcin and bone-specific phosphate alkaline collagen phosphatase were used to determine the functional markers of bone cell formation, antioxidant tartrate alkaline phosphatase (tpacp5b), type I alkaline collagen combined with cross-linked cells N-Terminal peptide (NTX) and human type I basic collagen combined with cross-linked cell C-Terminal Peptide (CTX) measure high levels of bone immune resorption function markers. Using an electric transfer device, the active protein that has been separated in the degreasing gel is transferred to a hydroxymethyl protein cellulose cell membrane by transfer, and the location of the non-specific active protein conjugate is blocked with 10 % defatted placenta and bovine protein serotonin. Nuclear metabolic factors are the main measurement markers for determining the metabolic pathways of bone nuclear metabolic factors Receptor activator of NF-kB (RANK)/ RANKL/OPG.
Statistical methods:
After the data in this study are verified to be correct, a metrology database is established, and the data in the metrology database is expressed with an accuracy x±s angle. Using p or t method to test, the count result data is expressed as a percentage coefficient (%) formula, p<0.05 as the main difference value has important statistical research significance. SPSS can be used to verify the normality analysis of variance between multiple groups of data. When there is no absolute mean between the multiple groups, the Trial of Prevention Strategies (TOPS) statistical method is used to compare the data. When the results are compared, the variance Chi-square (χ2) can be used.
Results and Discussion
During the experiment, none of the rats in the sham operation control group, ovariectomy control group and quercetin observation group died. There were no significant changes in the general behaviors of rats in the sham operation control group, ovariectomy control group, and quercetin observation group in daily diet, water intake, and body weight (p>0.05). After the experiment, the rats in the castration control group and the quercetin observation group felt sensitive and powerful, and their hair was shiny and moist. The rats in the sham-operated control group were unresponsive and had dark hair. After the experiment, the blood calcium and phosphorus levels and the blood magnesium level of the quercetin observation group increased. Compared with before the experiment, the difference was significant (p<0.05). There was no significant difference in the content of blood calcium, blood phosphorus, and blood magnesium between the quercetin observation group and that before the experiment (p>0.05). As shown in Table 2, the comparison results of serum indexes of rat bone tissue after quercetin intervention.
| Group | Blood calcium | Blood magnesium | Blood phosphorus | Lipids |
|---|---|---|---|---|
| Control | 44.25±4.235 | 55.63±4.11 | 78.61±5.42 | 95.22±6.39 |
| Castration | 38.54±3.66 | 62.35±4.36 | 81.63±5.08 | 98.83±6.82 |
| Observation | 39.57±3.47 | 69.47±3.95 | 74.46±4.95 | 120.6±7.47 |
| p | <0.05 | <0.05 | >0.05 | >0.05 |
Table 2: Comparison of Serum Indexes of RAT Bone Tissue after Quercetin Intervention
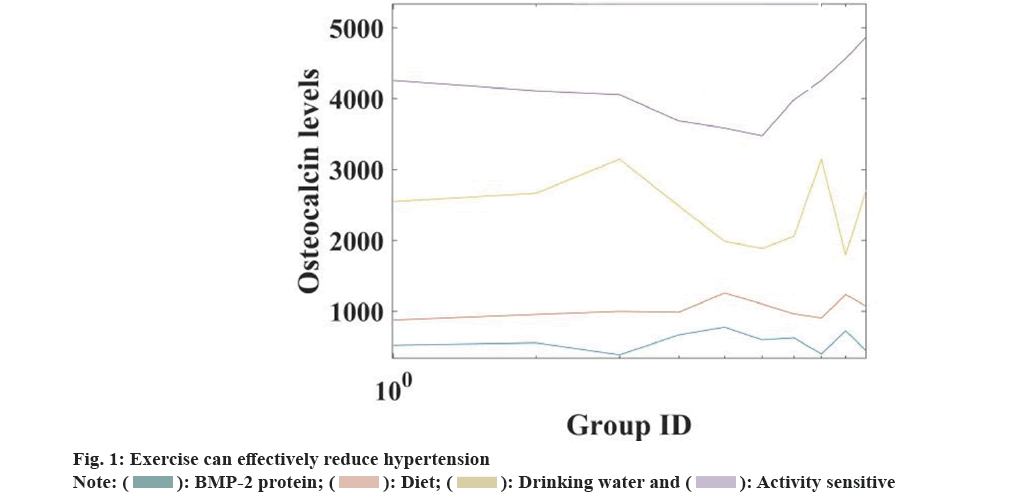
The results of the study showed that the expression of Bone Morphogenetic Protein-2 (BMP-2) protein in the femur of the ovariectomized control group and the quercetin observation group were changed. Compared with the sham operation control group, the BMP-2 protein concentration in the quercetin observation group was significantly increased (p<0.05). Compared with the sham operation control group, the difference between the ovariectomy control group and the quercetin observation group was statistically significant (p<0.05), suggesting that quercetin could significantly increase the BMP-2 protein expression in the femur of the rat, compared with the ovariectomy control group. Compared with the group, the expression of BMP-2 protein in the quercetin observation group was statistically significant (p<0.05), indicating that quercetin had a greater effect on BMP-2 protein concentration than ovariectomy. The results showed that there were statistically significant differences in the expression levels of type I collagen in the metaphysis of femoral shaft in the sham operation control group, ovariectomy control group and quercetin observation group. The expression level of type I collagen in femoral control group and quercetin observation group was higher than that in sham operation control group. Compared with the observation group, the difference was not statistically significant (p=0.086). The ovarian control group was lower than the quercetin observation group. The serum osteocalcin levels in the ovariectomized control group and the quercetin observation group were higher than those in the sham operation control group (p=0.004). There was no statistically significant difference between the quercetin observation group and the ovariectomized control group (p=0.761). The results of the study show that the serum osteocalcin levels of osteoporotic rats will be increased under the intervention of quercetin. The relevant data is shown in fig. 1.
From the data in fig. 1, it can be seen that the serum osteocalcin level of osteoporotic rats will be increased under the intervention of quercetin. The serum osteocalcin level has increased from the original (16.28±0.56) μg/l to (37.64±2.35) μg/l.
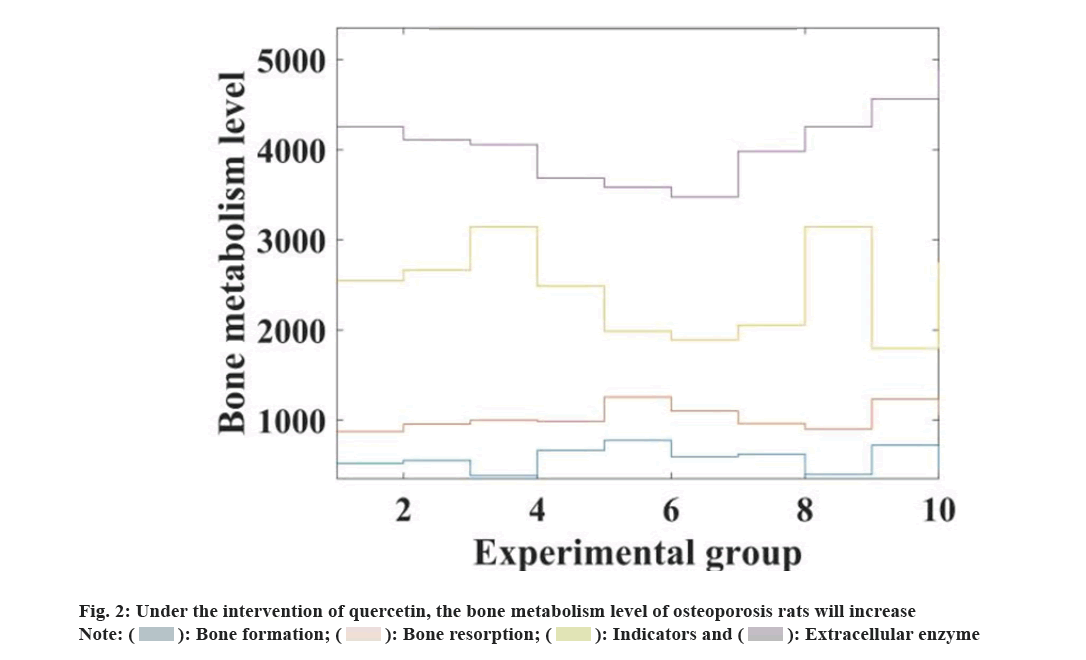
After 3 w and 5 w of quercetin intervention, bone metabolic levels of rats in each group were measured, and serum Bone Gla Protein (BGP), Bone-Specific Alkaline Phosphatase (BALP), Estradiol (E2) levels of osteoporosis rats were detected. The increasing trend of bone metabolism in model rats was positively correlated with the dose of quercetin. Compared with the ovariectomized group, the bone metabolism in the quercetin observation group was significantly increased at 5 w compared to 3 w (p<0.05), and there was no significant change in the ovarian control group. Compared with the sham operation control group, the medication was taken for 3 w at 5 w, the bone metabolism level of the quercetin observation group was further improved (p<0.05). Compared with the sham operation group, the ALP and osteocalcin of the ovariectomy control group and the quercetin observation group were significantly reduced (p<0.05), and Tartrate Resistant Acid Phosphatase (TRACP) was significant. The research results show that the bone metabolism level of osteoporotic rats will be increased under the intervention of quercetin, and the relevant data is shown in fig. 2.
From the data in fig. 2, it can be seen that the bone metabolism level of osteoporosis rats will be increased under the intervention of quercetin, and the bone metabolism index of osteoporosis rats will increase from the original (53.49±3.41) to (86.27.2±4.22).
Quercetin and its main derivative flavonoids are currently the most widely used polyhydroxy flavonoid organic compounds in nature. They also have many different biological activities. Not only can they effectively help prevent cell oxidation and eliminate free radicals in the body, but also have a variety of anti-cancer, anti-inflammatory, antibacterial, anti-malignant viruses, blood sugar and auxiliary blood pressure lowering, immune function regulation and promotion of cardiovascular health protection, etc. The researchers found that due to the continuous decrease of estrogen secretion in the body, human bones are in a new pathological state, and the conversion rate of bones in the early stage of osteoporosis will continue to increase, which can promote the formation of bones and white matter in the body. At the same time, the number of absorption increases, which further greatly aggravates the prevention and treatment of chronic osteoporosis. Therefore, the research in this paper explores the pharmacological effects and mechanism of serum quercetin on chronic osteoporosis by selecting and analyzing two serum osteocalcin metabolic marker hormones BGP and two serum osteocalcin. After 5 w of medication, the serum hormone ball in the sham operation group also increased slightly (p<0.05). As a kind of extracellular enzyme that inhibits osteoblasts, its high hormone content can directly reflect the activity of inhibiting osteoblasts, which also fully shows that the use of quercetin kinase can be affected at an early stage to inhibit osteoblasts. Activity, and the osteogenesis time continues to play a role in promoting the inhibition of osteogenic enzymes. Other clinical indicators of osteocalcin metabolism and human serum osteocalcin showed no significant changes in the early (3 w) cycle, but increased significantly at 5 w (p<0.05). Serum osteocalcin is not only a peptide chemical that directly reflects the transformation of quercetin phosphate bone cells under the action of calcium, but also because it has the effect of effectively promoting the deposition of calcium salt cells in bones and improving the success rate of bone calcium mineralization in bone cells. The characteristics can directly and effectively reverse the bone replacement rate or the success rate of bone cell transformation. The results show that quercetin can promote the accumulation of bone calcium in rats, increase the mineralization rate of bone cells, and accelerate the rate of bone formation. The relevant data is shown in fig. 3.
From the data in fig. 3, it can be seen that quercetin can promote the accumulation of bone calcium salts, increase the mineralization rate of bone cells, accelerate the rate of bone formation in rats, and increase the rate of bone calcium salt deposition by 18.7 %. And the mineralization rate increased by 22.6 % and the rat bone formation rate increased by 29.5 %.
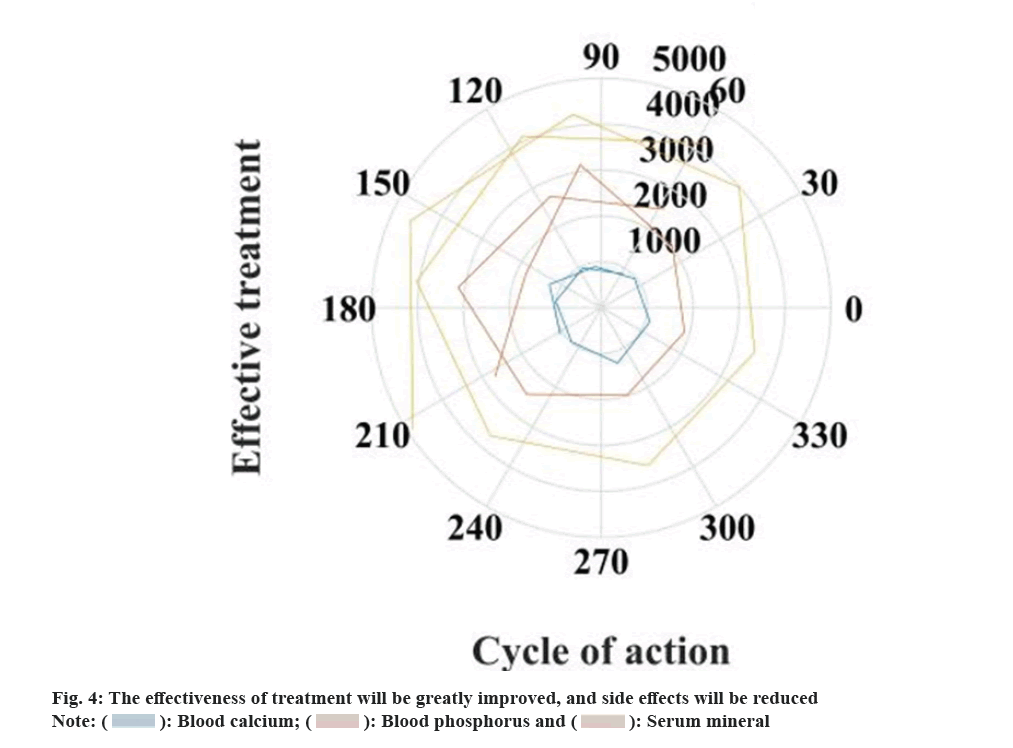
Osteoporosis leads to fractures due to the loss of osteoclast number and activity, resulting in a significant increase in the number of individual osteoclasts and active cells, resulting in a greatly increased elastic bone resorption rate, destruction, disappearance of trabecular bone tissue, subperiosteal. Cortical bone tissue destruction, mild elastic fracture of stress, secondary calcified osteophyte bone hyperplasia and other mechanical bone injuries. Bone tissue deformation severely compresses the bone nerves, increasing athletes with bone calcium deficiency, spinal activity load follows gravity. Changes in and curves greatly increase the activity load of the local muscles around the human spine. Quercetin achieves balance and regulation by affecting bone metabolism and serum osteocalcin, helping to treat chronic osteoporosis. At the same time, quercetin can also directly inhibit the effect of other substances on osteoclasts. Quercetin promotes the precise function of osteoclasts and endocrine, many osteoclast metabolic factors directly inhibits other osteoclast receptor cells. Only in this way can we give full play to the dual compound effect of cells promoting bone formation and bone breaking. Quercetin is different from the previous bone resorption calcium suppression supplements. It can increase bone mass and improve bone structure by up-regulating rat bone metabolism and serum osteocalcin expression level. Quercetin cannot only effectively increase the tissue thickness of trabecular bone and interosseous cortex, and the intensity of mechanical movement within the bone, but also effectively increase the area and bone density of bilateral lumbar spine in elderly postmenopausal women with osteoporosis, and improve the transformation of cortical bone capability index. The results of the study show that quercetin treatment of osteoporosis in rats can greatly improve the treatment efficiency and reduce side effects. The relevant data is shown in fig. 4.
It can be seen from fig. 4 that using quercetin drugs to treat osteoporosis in rats, the treatment efficiency will be increased by 25.2 %, and the side effects of drug treatment will be reduced by 34.8 %.
As my country has gradually entered an aging society, osteoporosis has become a common disease. This disease is mainly caused by the degeneration of bone tissue structure, which is easy to cause fractures and brings great problems to the lives of elderly patients. Quercetin is a flavonoid drug that is commonly used to treat osteoporosis. It has been clinically proven to have a good therapeutic effect and is worthy of further promotion and application.
The results of the study showed that under the intervention of quercetin, the bone metabolism index of osteoporosis rats increased from the original (53.49±3.41) to (86.27.2±4.22), and the serum calcium level from the original (16.28±0.56) µg/l increased to (37.64±2.35) μg/l. In addition, the bone density of the rats and the old tissue structure were also improved accordingly. It can be seen that quercetin can promote bone metabolism in osteoporosis rats and increase the content of serum osteocalcin in rats, and has a good effect on the prevention and treatment of osteoporosis diseases.
Studies have shown that quercetin can promote the accumulation of bone calcium, increase the mineralization rate of bone cells, accelerate the rate of bone formation in rats, increase the rate of bone calcium deposition by 18.7 %, and increase the mineralization rate of bone cells 22.6 %, rat bone formation rate increased by 29.5 %. Using quercetin drugs to treat osteoporosis in rats, the treatment efficiency will increase by 25.2 %, and the side effects of drug treatment will be reduced by 34.8 %.
Conflict of interests:
The authors declared no conflict of interests.
References
- Kanda J, Izumo N, Kobayashi Y, Onodera K, Shimakura T, Yamamoto N, et al. Effects of the antiepileptic drugs topiramate and lamotrigine on bone metabolism in rats. Biomed Res 2017;38(5):297-305.
[Crossref] [Google Scholar] [PubMed]
- Yu Z, Fan L, Li J, Ge Z, Dang X, Wang K. Lithium chloride attenuates the abnormal osteogenic/adipogenic differentiation of bone marrow-derived mesenchymal stem cells obtained from rats with steroid-related osteonecrosis by activating the β-catenin pathway. Int J Mol Med 2015;36(5):1264-72.
[Crossref] [Google Scholar] [PubMed]
- Oh KC, Hwang W, Park YB, Lee JH, Moon HS, Kim JH. Effects of alendronate on bone remodeling around osseointegrated implants in rats. Implant Dent 2017;26(1):46-53.
[Crossref] [Google Scholar] [PubMed]
- Jo YY, Seo SD, Kim JW, Cho HJ, Chon JW, Lee KG, et al. The effects of Cudrania tricuspidata extract on bone metabolism in ovariectomized rats. Int J Ind Entomol 2016;32(2):80-9.
- Oh KC, Moon HS, Lee JH, Park YB, Kim JH. Effects of alendronate on the peri-implant bone in rats. Oral Dis 2015;21(2):248-56.
[Crossref] [Google Scholar] [PubMed]
- Qu T, Zheng P, Yang C, Lan X, Zhang T, Liu H, et al. Effects of Danshensu on bone formation in ovariectomized rats. Zhejiang Da Xue Xue Bao Yi Xue Ban 2016;45(6):587-91.
[Crossref] [Google Scholar] [PubMed]
- Wu J, Cai XH, Qin XX, Liu YX. The effects of sclerostin antibody plus parathyroid hormone (1-34) on bone formation in ovariectomized rats. Z Gerontol Geriatr 2018;51(5):550-6.
[Crossref] [Google Scholar] [PubMed]
- Jeong JH, Park J, Jin ES, Min J, Jeon SR, Kim DK, et al. Adipose tissue-derived stem cells in the ovariectomy-induced postmenopausal osteoporosis rat model. Tissue Eng Regen Med 2015;12:28-36.
- Tezval M, Hansen S, Schmelz U, Komrakova M, Stuermer KM, Sehmisch S. Effect of urocortin on strength and microarchitecture of osteopenic rat femur. J Bone Miner Metab 2015;33:154-60.
[Crossref] [Google Scholar] [PubMed]
- Benson T, Menezes T, Campbell J, Bice A, Hood B, Prisby R. Mechanisms of vasodilation to PTH 1–84, PTH 1–34, and PTHrP 1–34 in rat bone resistance arteries. Osteoporos Int 2016;27(5):1817-26.
[Crossref] [Google Scholar] [PubMed]
- Liu YP, Ding XF, Huang MM, Chen Y, Peng J. Absorption and metabolism of icariin in different osteoporosis rat models. Zhongguo Zhong Yao Za Zhi 2016;41(13):2532-7.
[Google Scholar] [PubMed]
- Liu C, Zhang Y, Fu T, Liu Y, Wei S, Yang Y, et al. Effects of electromagnetic fields on bone loss in hyperthyroidism rat model. Bioelectromagnetics 2017;38(2):137-50.
[Crossref] [Google Scholar] [PubMed]