- Corresponding Author:
- A. Porel
East India Pharmaceutical Works Ltd., Quality Control Department, 119, Biren Roy Road (W), Kolkata - 700 0061, India
E-mail: abhijit_porel@mailcity.com
| Date of Submission | 26 June 2010 |
| Date of Revision | 22 February 2011 |
| Date of Acceptance | 25 February 2011 |
| Indian J Pharm Sci, 2011, 73 (1): 46-56 |
Abstract
The aim of the present study was the development and subsequent validation of a simple, precise and stability-indicating reversed phase HPLC method for the simultaneous determination of guaifenesin, terbutaline sulphate and bromhexine hydrochloride in the presence of their potential impurities in a single run. The photolytic as well as hydrolytic impurities were detected as 3,5-dihydroxybenzoic acid, 3,5-dihydroxybenzaldehyde, 1-(3,5-dihydroxyphenyl)-2- [(1,1-dimethylethyl) amino]-ethanone from terbutaline, 2-methoxyphenol and an unknown impurity identified as (2RS)-3-(2-hydroxyphenoxy)-propane-1,2-diol from guaifenesin. The chromatographic separation of all the three active components and their impurities was achieved on Wakosil II column, using phosphate buffer (pH 3.0) and acetonitrile as mobile phase which was delivered initially in the ratio of 80:20 (v/v) for 18 min, then changed to 60:40 (v/v) for next 12 min, and finally equilibrated back to 80:20 (v/v) for 10 min. Other HPLC parameters were: Flow rate at 1.0 ml/min, detection wavelengths 248 and 280 nm, injection volume 10 ml. The calibration graphs plotted with five concentrations of each component were linear with a regression coefficient R2 >0.9999. The limit of detection and limit of quantitation were estimated for all the five impurities. The established method was then validated for linearity, precision, accuracy, and specificity and demonstrated to be applicable to the determination of the active ingredients in commercial and model cough syrup. No interference from the formulation excipients was observed. These results suggest that this LC method can be used for the determination of multiple active ingredients and their impurities in a cough and cold syrup.
Keywords
Bromhexine hydrochloride, degradation products, guaifenesin, stability-indicating method, terbutaline sulfate, validation
Many pharmaceutical formulations against the common cold and cough syrup used to contain a combination of a bronchodialator terbutaline sulphate (TBN), mucolytic agent bromhexine hydrochloride (BHN) and an expectorant guaifenesin (GFN). They are chemically known as bis [(1RS)-(3,5- dihydroxyphenyl)-2- [(1,1-dimethylethyl)amino] ethanol]sulphate, N-(2-amino-3,5- dibromobenzyl)-Nmethylcyclohexanamine hydrochloride, and (2RS)-3- (2-methoxyphenoxy)-propane-1,2-diol, respectively. TBN is a synthetic β2−adrenoceptor agonist and enhances mucocillary transport to help expectoration. It is used as a bronchodialator in the treatment of bronchial asthma. GFN stimulate the bronchial glands lining of airway to produce a thin secretion that lubricates any thick mucous and making it easier to expel with coughing. It is known to increase the volume and reduce the viscosity of tenacious sputum [1]. BHN is a mucous modifying drug helps to improve the flow properties of bronchial mucous and eases expectoration. The excipients used are preservative, sweeteners, acidulates, artificial coloring and flavoring agent which are present in different proportions in the formulations. The simultaneous determination of the three components having different physicochemical properties is difficult and becomes complicated due to the matrix effect of inactive ingredients and with the presence of impurities.
To establish the stability characteristics, degradation studies (stress studies) were carried out on these three active ingredients under heat, water, acid, base, UV radiation and oxidative stress conditions, as recommended in the ICH guideline Q1A (R2) on stability testing of new drug substances and products [2].
A literature survey reveals some HPLC methods reported for the simultaneous determination of GFN [3-10] and BHN [11] along with some other active ingredients which exist as various combinations in cough-cold mixture and excipients in a multicomponent liquid dosage formulation as anticipated with the variation of mobile phase, column and detector. Some stability-indicating assay methods (SIAM) of GFN and TBN along with other active ingredients, excipients and impurities in liquid and solid dosage forms are reported [12-15]. Different HPLC methods for individual assay and related substances are available for TBN, GFN and BHN in official pharmacopoeia [16-18]. A few related impurities of TBN, BHN and GFN were mentioned in British Pharmacopoeia [17,18]. A few degradation products of TBN and a LC separation method were mentioned by Ahuja [19].
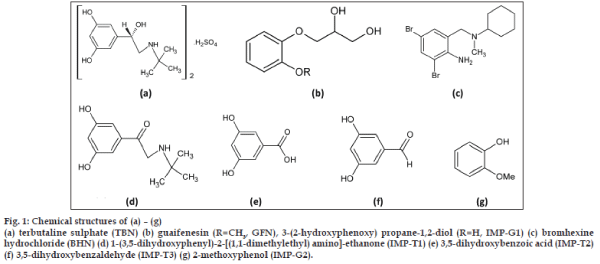
From the detailed literature survey it was found that no stability-indicating assay method reported yet for this combination of liquid dosage form, developed using the ICH approach of stress testing. A major demethylated photoproduct (2RS)-3-(2- hydroxyphenoxy)-propane-1,2-diol (IMP G), from GFN was not reported elsewhere. The structure of this impurity was confirmed by spectral (IR, 1H NMR, MS) studies and derivatizing into its acetate. The developed analytical procedure was validated to justify the suitability for the detection and quantification of degradation products as emphasizes in the ICH guideline Q3B (R2) entitled “Impurities in new drug products” [20]. Furthermore the method has been tested for liquid formulation.
Materials and Methods
Terbutaline sulphate was purchased from Neuland Laboratories Limited, India, bromhexine hydrochloride was purchased from Ven Petrochem Pvt. Ltd., India and guaifenesin was purchased from Granules India Limited, India. 1-(3,5-dihydroxyphenyl)-2- [(1,1-dimethylethyl)amino]-ethanone (IMP-T1) was purchased from Toronto Research Chemicals Inc. Canada; 3,5-dihydroxybenzoic acid (IMP-T2) and 3,5-dihydroxybenzaldehyde (IMP-T3) were obtained from Aldrich, Germany. (2RS)-3- (2-hydroxyphenoxy)- propane-1, 2-diol (IMP-G1) was synthesized in our laboratory; 2-methoxyphenol (IMP-G2) was obtained from Fluka, Germany. High purity water was prepared using Milli Q purification system from Millipore (Peenya, Bangalore, India).
Photo degradation was carried out in a photostability chamber (Thermolab, India) equipped with a light bank consisting of two UV (TUV 15W) and four fluorescent (TLD 15W) lamps. The HPLC system consisted of waters 1525 binary pump, waters 717 plus auto sampler, a waters 2487 dual λ absorbance detector (Waters, Milford, MA, USA). Acquisition of Chromatographic data was made with Empower version 2.0 software. The chromatographic separations were achieved on a Wakosil II C18 (150×4.6 mm, i.d., 5μ) column from Waters Corporation. IR experiments were performed with a FT-IR spectrometer (Perkin- Elmer Spectrum 2000, Wellesley, M.A. USA). NMR experiments were carried on a NMR spectrometer (BRUKER 300 MHz, Milton, Ontario, Canada). The proton chemical shifts were referenced to the TMS signal at 0 ppm. Electrospray mass spectrometry measurements were performed on a MS-TOF micromass spectrometer (Waters, USA).
Optimized chromatographic conditions
HPLC studies were carried out on all the reaction solutions individually and on a mixture of the solutions in which decomposition was observed. The separations were achieved by gradient elution using phosphate buffer (25 mM disodium hydrogen phosphate dodecahydrate and 25 mM potassium dihydrogen orthophosphate, pH 3.0) and acetonitrile as the mobile phase. The phosphate buffer was prepared by dissolving 8.95 g of disodium hydrogen phosphate dodecahydrate and 3.40 g of potassium dihydrogen orthophosphate in 1000 ml water and final pH was adjusted by adding orthophosphoric acid and 2N sodium hydroxide. It was filtered through 0.45 μm nylon filter and degassed before use. The diluting solvent was prepared by mixing the same phosphate buffer (after raising pH to 3.5) with acetonitrile in the ratio of 80:20. The injection volume was 10 μl and mobile phase flow rate was 1.0 ml/min. The detection was carried out at 248 nm for BHN and 280 nm for TBN and GFN.
Preparation of stock and standard solutions
For the preparation of stock solutions, 100 mg each of terbutaline sulphate, bromhexine hydrochloride and guaifenesin working standard (WS) was taken in a 100 ml volumetric flask and it was made up to volume with the diluting solvent (Stock solution A). For the calibration of the assay method, 0.1, 0.5, 1.0, 2.0 and 5.0 ml aliquots of the stock solution A were diluted to 10 ml with the diluting solvent to give the final concentrations of 10, 50, 100, 200 and 500 μg/ ml.
Calibration solutions for the impurity method were prepared containing each of the related compounds at concentration ranging 0.5 μg/ml to 20.0 μg/ml (0.5, 1.0, 5.0, 10.0 and 20.0 μg/ml) and the active ingredients at the concentrations ranging from 10 μg/ ml to 500 μg/ml.
Preparation of sample solution
Accurately measured volumes of the cough syrup equivalent to 10 mg each of BHN, TBN and GFN was taken in a 50 ml volumetric flask and made up the volume with diluting solvent. Final solution had a concentration of 200 μg/ml of each component.
Specificity and stress study
Specificity is the ability of the method to measure the analyte response in the presence of its potential impurities. The specificity of the LC method developed for TBN, BHN and GFN was determined in the presence of their impurities, namely IMP-T1, IMP-T2, IMP-T3, IMP-G1 and IMP-G2. Stress studies were performed for TBN, BHN and GFN in bulk drug individually and in synthetic mixtures to assess the suitability of the method as stabilityindicator. Forced degradation under stress conditions of UV light (254 nm), heat (70°), acid (0.1N HCl), base (0.1 NaOH) and oxidation (3.0% H2O2) was used to evaluate the ability of the proposed method to separate three active ingredients (AI) from the degradation products. All degradation experiments on TBN, BHN and GFN were performed at a drug concentration of 500 μg/ml.
Linearity
Linearity test solutions for the assay method and for the related substance method were prepared at five concentration levels in the range of 10 μg/ml to 500 μg/ml and from 0.25% to 10.0% of target analyte concentration (TAC), 200 μg/ml (0.5, 1.0, 5.0, 10.0 and 20.0 μg/ml), respectively. The peak area versus concentration data were analyzed by least square linear regression.
Precision
Assay method precision was evaluated by carrying out six independent assays of test sample of TBN, BHN and GFN at 100% level of the test concentration, 200 μg/ml. The precision of the related substance method was checked by injecting six individual preparations of TBN, BHN and GFN (200 μg/ml), spiked with 2.5% each of IMP-T1, IMP-T2, IMP-T3, IMP-G1 and IMP-G2. The % RSD of the area for each impurity was calculated. The intermediate precision of the method was also evaluated using different analyst and different instruments in the same laboratory.
Accuracy
The accuracy of the assay method was evaluated in triplicate at three different concentration levels of every compound, 100, 200 and 400 μg/ml (50%, 100% and 200% of the analyte concentration). The accuracy study for impurities was carried out in triplicate at 0.25%, 0.5% and 1.0% of the TBN, BHN and GFN analyte concentrations, (200 μg/ml). The percentage recovery at each level was determined by comparison to the known amount added.
Limit of detection and limit of quantitation
The limit of detection (LOD) and limit of quantitation (LOQ) were determined by the slope method by injecting a series of dilute solutions with known concentrations. A precision study was also carried at the LOQ level by injecting six individual preparations of IMP-T1, IMP-T2, IMP-T3, IMP-G1 and IMP-G2 and calculating the % RSD of the area.
Robustness
To determine the robustness of the method developed, the experimental conditions were deliberately altered and the chromatographic parameters viz., capacity factor (K'), tailing factor (T), no. of theoretical plates (N) and percent recoveries were recorded. The flow rate of the mobile phase was 1.0 ml/min. To study the effect of flow rate on the resolution, 0.05 units flow was changed, from 0.95 to 1.05 ml/min. The effect of mobile phase was studied by varying acetonitrile from –10% to +10% and the pH of the mobile phase was studied by varying pH by –0.12 to +0.12, while other mobile phase components were held constant.
Solution stability and mobile phase stability
The solution stability of TBN, BHN and GFN and their impurities in the related substance method was investigated by leaving sample solutions in tightly capped volumetric flask at room temperature for 72 h. The contents of impurities were determined every 24 h up to the study period. The mobile phase stability was also investigated for 48 h by injecting the freshly prepared solutions every 6 h. Contents of impurities and active ingredients were checked in the test solutions.
Results and Discussion
The main purpose of the chromatographic method was to separate IMP-T1, IMP-T2, IMP-T3, IMP-G1, IMP-G2 (fig. 1) and the degradation products generated during stress studies from the three active ingredient peaks. To achieve better separation of all the analytes in the chromatogram several trials were done with the variation of column, composition of mobile phase and diluting solvent, pH of the buffer, mode of elution either with isocratic or gradient. The results are summarized in Table 1. Octadecylsilyl silica gel column with different mobile phase is recommended for the determination of individual assay and related substance of TBN, BHN and GFN in different international pharmacopoeia [16,17]. Two different types of octadecylsilyl silica gel columns: Atlantis d C18 (150×4.6 mm, 3 μm) and Wakosil II C18 (150×4.6 mm, 3 μm), were compared on the basis of retention factor of very neighbouring component peaks to evaluate the column performance. A polar column Hypersil silica (250×4.6 mm, 3 μm) was also tried with. Results of this comparison showed that the Wakosil II column was the most suitable for the separation of three active components and their potential impurities in a single run and this column was used for all other experiments. By employing Wakosil II C18 column (150×4.6 mm, 3 μm), mobile phase with a phosphate buffer and acetonitrile (60:40, v/v) enabled to elute three active components with bad resolution between GFN and TBN (exp no. 8). Increasing the phosphate buffer proportion to 80% reasonable resolution between GFN and TBN was achieved with long retention of BHN (exp no. 9). To compromise these differences of elution in isocratic mode a need for gradient elution mode was felt. Due to the serious differences in polarity of the active ingredients, the pH of a chosen mobile phase and the diluting solvent had a significant influence on separation (exp. no 10 and 11). The optimal gradient selected was the following: First a linear gradient to solvent A, (phosphate buffer, pH 3.0) – Solvent B, (acetonitrile) (80:20, v/v) mixture during 18 min, then a second linear gradient to solvent A – solvent B (60:40, v/v) during 12 min. Finally, the initial conditions were established in 10 min.
Fig. 1: Chemical structures of (a) – (g) (a) terbutaline sulphate (TBN) (b) guaifenesin (R=CH3, GFN), 3-(2-hydroxyphenoxy) propane-1,2-diol (R=H, IMP-G1) (c) bromhexine hydrochloride (BHN) (d) 1-(3,5-dihydroxyphenyl)-2- [(1,1-dimethylethyl) amino]-ethanone (IMP-T1) (e) 3,5-dihydroxybenzoic acid (IMP-T2) (f) 3,5-dihydroxybenzaldehyde (IMP-T3) (g) 2-methoxyphenol (IMP-G2).
| Mobile phasecomponent | pH | Column | Diluting solvent | Flow rate(ml/min) | Remarks | ||
|---|---|---|---|---|---|---|---|
| Water:buffera:acetonitrile | 6.51 | Hypersil silica | Mobile phase | 0.7 | No separation between GFN and IMP-G2 | ||
| (25:50:25 v/v) | (250×4.6 mm; 5 µm) | and fast elution of both the components | |||||
| Acetic acid (0.1%v/v):acetonitrile | 6.51 | Hypersil silica | Mobile phase | 0.7 | High tailing factor for BHN and bad | ||
| (40:60, v/v) (pH adjusted with | (250×4.6 mm; 5 µm) | resolution between GFN and TBN. | |||||
| TEA) | |||||||
| Acetic acid (0.1%v/v): | 6.51 | Atlantis dC18, | Mobile phase | 1.5 | Bad resolution between the peaks TBN | ||
| acetonitrile (55:45) (pH adjusted | (150×4.6 mm; 4 µm) | and BHN. | |||||
| with TEA) | |||||||
| Bufferb: acetonitrile: methanol | 4.01 | Atlantis dC , 18 (150×4.6 mm; 4µm) | Mobile phase | 1.0 | Bad resolution between GFN and TBN, no | ||
| (20:20:60,v/v) | 4.22 | elution for BHN | |||||
| Bufferb:acetonitrile (80:20,v/v) | 4.01 | Atlantis dC , 18 (150×4.6 mm; 4 µm) | Mobile phase | 1.0 | Fast elution of GFN and TBN, bad | ||
| 4.22 | resolution between the the peaks of GFN | ||||||
| Acetic acid (1% v/v):acetonitrile (80:20,v/v) |
3.41 | Atlantis dC18, (150×4.6 mm; 4 μm) |
Mobile phase | 1.0 | Bad resolution between TBN and IMP-T1, no elution for BHN |
||
| Phosphoric acid (0.05%v/v):acetonitrile (20:80,v/v) |
7.01 | Atlantis dC18, (150×4.6 mm; 4 μm) |
Methanol | 2.0 | No elution for GFN and TBN | ||
| Bufferb:acetonitrile (60:40,v/v) | 4.01 | Wakosil II C18 | Mobile phase | 1.0 | Bad resolution between GFN and TBN peaks |
||
| Bufferb:acetonitrile (80:20,v/v) | 4.01 | Wakosil II C18 | Mobile phase | 1.0 | Long retention time for BHN peak | ||
| 4.22 | (150×4.6 mm; 3 μm) | ||||||
| Bufferb:acetonitrile (gradient*) | 4.01 | Wakosil II C18 (150×4.6 mm; 3 μm) |
ACN:bufferb (20:80,v/v, pH 4.0) |
1.0 | Bad resolution among TBN, IMP-T2 and IMP-T1 peaks |
||
| Bufferb:acetonitrile (gradient*) | 3.01 | Wakosil II C18 (150×4.6 mm; 3 μm) |
ACN:bufferb (20:80,v/v, pH 3.5) |
1.0 | Acceptable retention time for all peaks. Resolution and peak characteristics were good. |
||
1pH of Buffer; 2pH of Mobile phase; aBuffer of pH 6 prepared by dissolving 1.36 g KH2PO4 in 500 ml water and by adjusting pH by NaOH or H3PO4. bBuffer of pH 4 prepared by dissolving 8.95 g Na2HPO412H2O and 3.40 g KH2PO4 in 1000 ml water and by adjusting pH by NaOH or H3PO4;* Gradient: 0-18 min; Acetonitrile 20%; 19-30 min, Acetonitrile 20-40%; 31-40 min, Acetonitrile 40-20%.
Table 1: Method optimization data.
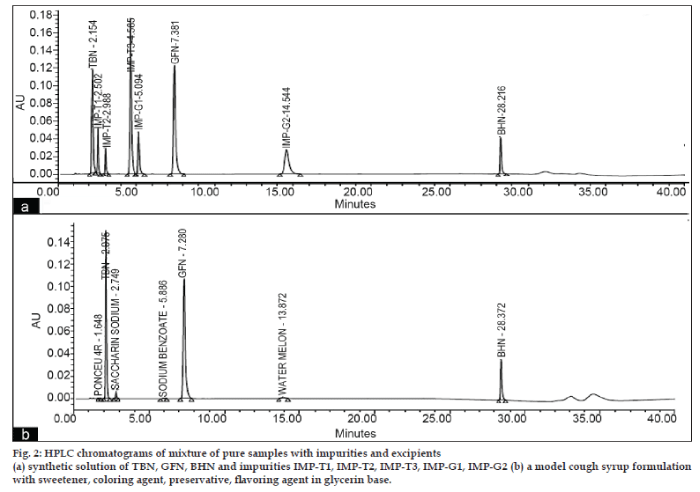
HPLC chromatograms in optimized chromatographic condition for mixtures of three active ingredients with all the five impurities (fig. 2a) and in a model cough syrup formulation (fig. 2b) showed that all the peaks were well separated from the immediate neighboring peak.
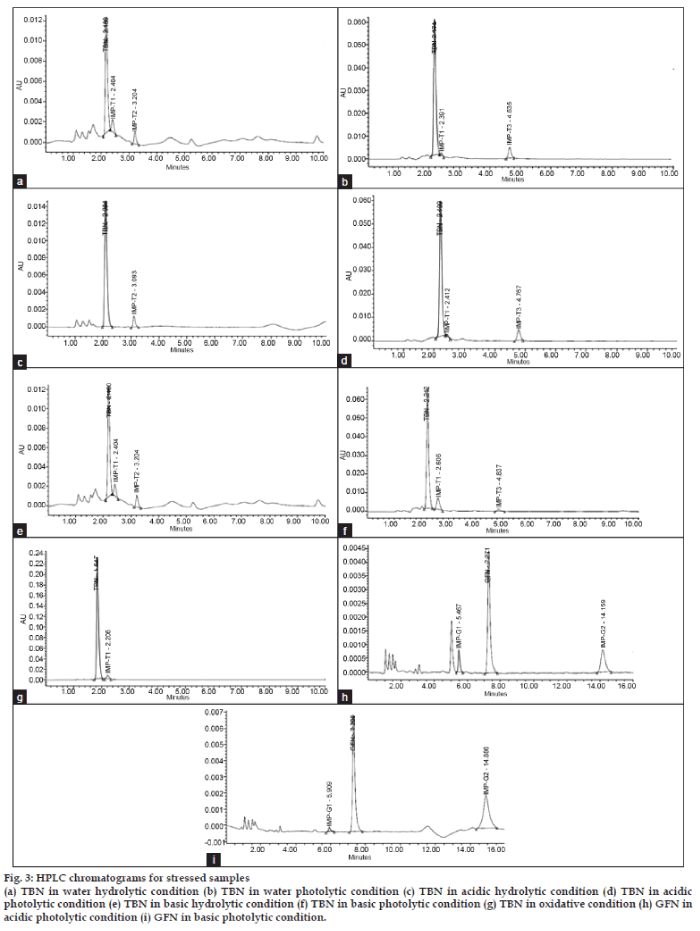
To assess the suitability of the method as a stability indicator, stress testing was performed. In most of the different stressed conditions of bulk drugs, moderate to high degradation was found. In GFN and TBN, some impurities among the probable five mentioned impurities were detected, and no impurity peak was detected in BHN even after complete degradation. In neutral and acidic photolytic conditions major formation of IMP-T3 was found in contrast to the IMP-T2 in hydrolytic conditions. The same trend of photolytic degradation of each ingredient was observed by mixing three components together in the model cough syrup. The detailed data are as summarized in Table 2. HPLC chromatograms for all the degradation studies are given in fig. 3.
| Stress condition | Time (h) Temp (°) |
% Degradation* | Degradation producta | ||||||
|---|---|---|---|---|---|---|---|---|---|
| GFN | BHN | TBN | GFN | TBN | |||||
| Relative retention | % | Relative retention | |||||||
| % | |||||||||
| Base hydrolysis (0.1 N aq, NaOH) | 20 | 8.79 | 98.13 | 84.62 | _ | _ | 1.1(IMP-T1) | 0.015 | |
| 80 | 1.97 | 0.01 | |||||||
| Acid hydrolysis | 20 | 10.90 | 10.90 | 77.75 | _ | _ | 1.47(IMP-T2) | 1.67 | |
| 80 | |||||||||
| Neutral hydrolysis (H2O) | 20 | 9.50 | 1.2 | 76.6 | _ | _ | 1.47(IMP-T2) | 12.56 | |
| 80 | |||||||||
| Photo degradation in H2O | 72 | 30 | 55.09 | 22.86 | _ | _ | 1.09 (IMP-T1) | 1.03 | |
| 40 | (41.68) | (79.4) | (23.91) | 2.13 (IMP-T3) | 8.3 | ||||
| Photo degradation in 0.1 N aq. HCl | 72 | 64 | 87.35 | 21.69 | 0.69 (IMP-G1) | 10.0 | 1.09 (IMP-T1) | 0.9 | |
| 40 | (43.68) | (64.96) | (39.47) | 0.75 | 5.0 | 2.13 (IMP-T3) | 8.27 | ||
| 1.94 (IMP-G2) | 15.0 | ||||||||
| Photo degradation in 0.1 N aq. | 72 | 55.85 | 100.0 | 80.00 | 0.69 (IMP-G1) | 0.5 | 1.17(IMP-T1) | 10.79 | |
| NaOH | 40 | (53.37) | (98.25) | (81.51) | 0.75 | 1.6 | 2.18(IMP-T3) | 1.41 | |
| 1.94 (IMP-G2) | 36.0 | ||||||||
| Oxidation in H2O2 | 20 | 25.68 | 7.0 | 10.5 | _ | _ | 1.1(IMP-T1) | 10.71 | |
| 80 | |||||||||
*Degradation in the mixture of the components is shown in bracket; aThe recovery of unknown degradation products was calculated using respective active ingredient (GFN and TBN) as a reference standard
Table 2: Summary of forced degradation results.
Fig. 3: HPLC chromatograms for stressed samples (a) TBN in water hydrolytic condition (b) TBN in water photolytic condition (c) TBN in acidic hydrolytic condition (d) TBN in acidic photolytic condition (e) TBN in basic hydrolytic condition (f) TBN in basic photolytic condition (g) TBN in oxidative condition (h) GFN in acidic photolytic condition (i) GFN in basic photolytic condition.
System suitability and system precision were performed daily by evaluating number of theoretical plates (N), USP resolution between neighboring peaks (Rs) and USP tailing factor (T), which were found well within the specified USP limit (Rs>2, N>4000 and T<1.5). The calibration curves were constructed covering both high level (10-500 μg/ ml) and low level (0.5-20.0 μg/ml) concentration for the purpose of active ingredient (AI) and impurity analysis in cough syrup and plotting concentration (μg/ml) against peak area. For each component, a series of five concentration points were prepared and each solution was injected six times. A summary of the data showing the slopes, y-intercept values and 95% Confidence Interval (C.I.) for the slope and y- intercept values for assay and impurities is given in Table 3. The correlation coefficient for the assay of active components and analysis of impurities were all greater than 0.999. In addition, the analysis of residuals for the assay and the impurity showed that the values are randomly scattered around zero, which shows a good fit with the linear model.
| Parameter | Compound | |||||||
|---|---|---|---|---|---|---|---|---|
| GFN | TBN | BHN | IMP-T1 | IMP-T2 | IMP-T3 | IMP-G1 | IMP-G2 | |
| Range (µg/ml) | 10-500 | 10-500 | 10 -500 | 0.5-20.0 | 0.5-20.0 | 0.5-20.0 | 0.5-20.0 | 0.5-20.0 |
| Slope | 0.520 | 0.409 | 0.142 | 0.1624 | 2.733 | 2.39 | 6.25 | 8.90 |
| 95% CI for mean slope | ±0.007 | ±0.01 | ±0.002 | ±0.0013 | ±0.002 | ±0.01 | ±0.06 | ±0.01 |
| Intercept | 4.913 | 2.470 | 1.142 | 0.05 | -0.61 | 1.52 | -1.04 | -3.76 |
| 95% CI for mean intercept | ±1.48 | ±0.99 | ±0.40 | ±0.02 | ±0.04 | ±0.43 | ±0.18 | ±0.27 |
Number of points on each regression line is five, TBN - bronchodialator, terbutaline sulphate; BHN - mucolytic agent, bromhexine hydrochloride; GFN - expectorant, guaifenesin
Table 3: Linearity and regression data
The intraday precision showed overall percent recoveries in the range of 98.68 to 99.62% with RSD ranging from 1.51 to 2.08% for the three active ingredients and overall percent recoveries 99.03 to 100.36 with RSD ranging from 0.38 to 1.40% for the impurities. The interday precision showed overall percent recoveries in the range of 99.99 to 102.14% with RSD ranging from 0.90 to 2.30% for the three active ingredients and overall percent recoveries 99.28 to 101.98 with RSD ranging from 0.10 to 2.30% for the impurities. The intermediate precision for analysis of active ingredients and impurities showed percent recoveries in the range of 97.75 to 101.50 with %RSD less than 2.0. By applying analysis of variance (ANOVA) test to the recovery results, the statistical results with 95% confidence limit indicate that there was no significant differences between inter and intra-day analysis results in respect of tabulated F value. The results are summarized in Table 4.
| Substance | Added conc. (µg/ml) | Repeatability | Intermediate precision | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Intra-day measured concentration | Inter-day measured concentration | Different analyst (day, column) measured concentration | ||||||||
| Recovery (%) | RSD (%) | Recovery (%) | RSD (%) | Recovery (%) | RSD (%) | |||||
| TBN | 200 | 98.68 | 1.51 | 100.78 | 0.9 | 101.5 | 1.3 | |||
| BHN | 200 | 98.91 | 1.79 | 99.99 | 1.3 | 101.68 | 1.5 | |||
| GFN | 200 | 99.62 | 2.08 | 102.14 | 2.3 | 101.18 | 1.3 | |||
Repeatability and intermediate precision data of HPLC assay of TBN, BHN and GFN (n=6), TBN - bronchodialator, terbutaline sulphate; BHN - mucolytic agent, bromhexine hydrochloride; GFN - expectorant, guaifenesin
Table 4: Repeatability and intermediate precision data
The accuracy for the applied HPLC method was confirmed by applying it to the synthetic mixtures of TBN, BHN, and GFN and their impurities in different concentration levels of 100, 200, 400 μg/ ml. Mean recovery of nine (3×3) replicates of GFN ranged from 100.10 to 100.83% with RSD range 0.30 to 0.80%, overall recovery 100.50% and a 95% confidence interval (CI) of ±0.21. Mean recovery of nine replicates of TBN ranged from 98.81 to 100.63% with RSD range 0.30 to 1.40%, overall recovery 99.66% and a 95% confidence interval (CI) of ±0.71. Mean recovery of nine replicates of BHN ranged from 98.80 to 101.15% with RSD range 0.80 to 1.56%, overall recovery 100.08% and a 95% confidence interval (CI) of ±0.91. The accuracy of the method for the impurity analysis was determined by fortifying placebo and three active drug substances with known amount of the five impurities at three different concentration levels ranging from 0.5 to 2.0 μg/ml. The mean recovery of impurities of three replicates remained within 93.00% to 100.5% and % RSD less than 5.0. This indicates that the assay value obtained accurately represented the true drug content in the formulation over the method range 10-500 μg/ml. No difference in precision between the upper and lower limits of the method ranges was found using an ANOVA at the 95% confidence level as evidenced by the F ratio for each component. The results are summarized in Table 5.
| Substances | Added (µg/ml) | Found (µg/ml) | Recovery (%) | RSD (%) |
|---|---|---|---|---|
| TBN | 100.00 | 98.81 | 98.81 | 0.40 |
| BHN | 100.00 | 101.15 | 101.15 | 0.80 |
| GFN | 100.00 | 100.58 | 100.58 | 0.30 |
| IMP T-1 | 0.50 | 0.515 | 103.00 | 2.30 |
| IMP T-2 | 0.50 | 0.499 | 99.80 | 2.40 |
| IMP T-3 | 0.50 | 0.492 | 98.40 | 0.40 |
| IMP G-1 | 0.50 | 0.513 | 102.60 | 0.60 |
| IMP G-2 | 0.50 | 0.500 | 100.00 | 1.30 |
| TBN | 200.00 | 201.26 | 100.63 | 1.40 |
| BHN | 200.00 | 197.76 | 98.80 | 1.56 |
| GFN | 200.00 | 201.67 | 100.83 | 0.40 |
| IMP T-1 | 1.00 | 0.93 | 93.00 | 4.60 |
| IMP T-2 | 1.00 | 1.00 | 100.00 | 0.70 |
| IMP T-3 | 1.00 | 1.00 | 100.00 | 0.50 |
| IMP G-1 | 1.00 | 0.99 | 99.74 | 0.20 |
| IMP G-2 | 1.00 | 1.00 | 100.00 | 0.50 |
| TBN | 400.00 | 398.22 | 99.55 | 0.3 |
| BHN | 400.00 | 401.16 | 100.29 | 1.52 |
| GFN | 400.00 | 400.4 | 100.10 | 0.80 |
| IMP T-1 | 2.00 | 1.99 | 99.55 | 4.20 |
| IMP T-2 | 2.00 | 2.01 | 100.50 | 0.80 |
| IMP T-3 | 2.00 | 1.98 | 99.15 | 0.90 |
| IMP G-1 | 2.00 | 2.00 | 100.00 | 1.40 |
| IMP G-2 | 2.00 | 2.00 | 100.00 | 0.70 |
Results of recovery analysis of TBN, GFN, BHN and its impurities at different concentration level (n=3), TBN - bronchodialator, terbutaline sulphate; BHN - mucolytic agent, bromhexine hydrochloride; GFN - expectorant, guaifenesin
Table 5: Recovery data.
The limit of detection (LOD) of IMP-T1, IMP-T2, IMP-T3, IMP-G1 and IMP-G2 were found to be 0.156, 0.033, 0.135, 0.061 and 0.025 μg/ml at a signal to noise ratio 3:1. The limits of quantitation (LOQ) of IMP-T1, IMP-T2, IMP-T3, IMP-G1 and IMP-G2 were found to be 0.483, 0.099, 0.411, 0.182 and 0.077 μg/ml. The precision of the impurities at LOQ level was below 5.0% RSD.
In the robustness study with the small but deliberate variations in method parameters e.g., pH of buffer in mobile phase (pH 3.12, 3.00, 2.88), mobile phase ratio (buffer: ACN, v/v 82:18, 80:20, 78:22) and flow rate (0.95, 1.00, 1.05 ml/min) there were no dramatic changes in the chromatographic parameters (K', T and N) for active ingredients found. Percent recoveries of three active ingredients remained within the range 98.69-101.81 with %RSD ranged between 0.1 to 2.0.
The RSD of the assay of all the three active ingredients namely TBN, BHN and GFN during solution stability and mobile phase stability study was within 1.40 % assuring that sample solutions and mobile phase used during assays can be preserved for 72 h and 48 h, respectively.
The applicability of the validated method was also tested by analyzing samples of a model cough syrup and a commercial cough syrup containing the three active ingredients and excipients namely sodium benzoate, saccharin sodium, ponceau 4R supra and water melon flavor in glycerin base. The chromatogram of the model cough syrup analysis is shown in fig. 2b. The recovery (99.94% to 100.01%) and %RSD (0.025 to 1.56) of the assay method were well within the limit and indicated that the proposed method can be used for quantitation of TBN, BHN and GFN and impurities in a single run, in routine quality control analysis of cough-cold syrup.
The Infrared spectrum of IMP-G1 revealed a sharp band at 3650 cm-1 in very dilute solution of carbon tetrachloride due to the presence of “free” phenolic hydroxyl group. The other characteristic bands in KBr are as follows (cm-1): 3800-3200 (O-H stretching mode associated with hydroxyl groups), 1920-1600 (ortho- substituted aromatic overtones), 1350-1263 (C=C stretching of aromatic group), 1266-1092 (C-O stretching vibration of phenolic and aliphatic hydroxyl group).
The assignments of signals of the NMR spectra of IMP-G1 and its acetate derivative (prepared by usual procedure of acetylation) are given as follows:
1H NMR of IMP-G1 (300 MHz, CDCI3): δ 6.79-6.94 (4H, m, Ar-H), 4.89 (1H, brs, Ar-OH), 4.10 (1H, d, J=6 Hz, hydroxyl proton of -CHOH-), 3.95-4.05 (3H, m, methylene and methine protons of Ar-OCH2- CHOH-), 3.70 (2H, d, J=4.5 Hz, methylene protons of –CH(OH)OCH2-), 3.32 (1H, s, hydroxyl proton of -CH2OH).
1H NMR of acetate of IMP-G1 (300 MHz, CDCl3): δ 6.84-7.12 (4H, m, Ar-H), 5.31 (1H, m, methine protons of Ar-OCH2-CHOH-), 4.28 (1H, dd, J1=12 Hz and J2=3.9 Hz, Ha proton of Ar-OCH2-), 4.14 (1H, dd, J1=12 Hz and J2=5.7 Hz, Hb proton of –ArOCH2-), 4.03 (2H, d, methylene protons of -CH2OH), 2.2, 2.0, 1.99 (3 × 3H, s, methyl protons of three -OCOCH3).
The presence of three hydroxyl groups in the IMP-G1 was confirmed by the appearance of three distinct acetate singlets at 2.2, 2.0 and 1.99 ppm in its acetate derivative. The absence of any signal for o-methoxy group further supports the demethylation leading to the formation of IMP-G1. The two geminal protons HaHb of the aromatic –OCH2 group are diastereotopic and so anisochronous, and showed two different chemical shift values. The equatorial Ha proton exhibited geminal coupling with axial Hb proton and also with the vicinal proton of –CHOH at 4.28 ppm (J1=12 Hz, J2=3.9 Hz) as AB quartet. The axial proton Hb coupled similarly with equatorial counterpart Ha and also with vicinal pseudoaxial proton of -CHOH at 4.14 ppm as AB quartet. The pseudoaxial proton of –CHOH group appeared as a multiplet at 5.31 ppm. The other two geminal protons of the –CH2OH group appeared as doublet at 4.03 ppm.
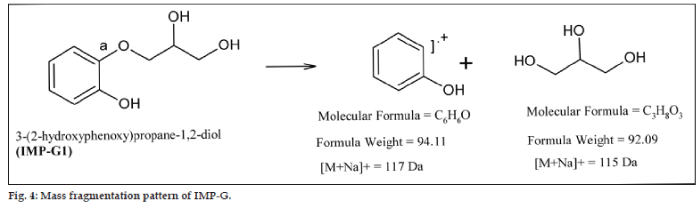
The positive ion Electrospray Ionization (ESI) mass spectrum of IMP-G1 exhibited a sodiated molecular ion, [M+Na]+ at m/z 207 confirming the molecular weight 184 Da. The decrease of 14 mass units from GFN molecular weight 198, suggested the demethylation of GFN. To obtain additional structural information, ESI- MS/MS analyses were performed. The MS/MS spectrum of IMP-G1 contained the sodiated product ions, m/z 115, 117 (100%), 145 (70%), 133 (20%), 99.9 (15%), 174 (20%) (rounded value given). Cleavage of bond (a) resulted in product ions at m/z 115 [M+Na]+, representing the aliphatic portion and m/z 117 [M+Na]+ representing the demythylated aromatic portion of the molecule (fig. 4). Therefore the MS/MS data confirmed the demethylated structure for IMP-G1. Based on the combination of the IR, NMR and MS data, IMP G-1 was identified as (2RS)-3-(2-hydroxyphenoxy)-propane-1,2-diol.
Acknowledgements
The authors would like to thank the management of East India Pharmaceutical Works Limited, Kolkata, India, for their support to carry out this investigation.
References
- Heiborn H, Regelow KO, Odeblad E. Effect of bromhexine and Guaiphenesin on clinical state, ventilatory capacity and sputum viscosity in chronic asthma. Scand J Respir Dis 1976;57:88-96.
- ICH, Q1A (R2), Stability Testing of New Drug Substances and Products, Harmonised Tripartite Guideline, 2003. p. 1-18.
- Schieffer GW, Hughes DE. Simultaneous stability-indicating determination of phenylephrine hydrochloride, phenylpropanolmine hydrochloride and guaiphenesin in dosage forms by reversed phase paired-ion high-performance liquid chromatography. J Pharm Sci 1983;72:55-9.
- Amer SM, Abbas SS, Shehata MA, Ali NM. Simultaneous determination of phenylephrine hydrochloride, guaiphenesin and chlorpheniramine maleate in cough syrup by gradient liquid chromatography. J AOAC Int 2008;91:276-84.
- Jain JK, Prokash MS, Mishra RK, Khandhar AP. Simultaneous determination of multi drug components Theophylline, Etofylline, Guaiphenesin and Ambroxol Hydrochloride by validated RP-HPLC method in liquid dosage form. Pak J Pharm Sci 2008;21:151-8.
- Chen TM, Panifico JR, Dalv RE. High-pressure liquid chromatographic assay of dextromethorphan hydrochloride, guaiphenesin and sodium benzoate in an expectorant syrup. J Chromatogr Sci 1988;26:636-9.
- Carnovale L. Simultaneous determination of acetaminophen, guaiphenesin, pseudoephedrine, pholcodine and paraben preservatives in cough mixture by high-performance liquid chromatography. J Pharm Sci 1983;72:196-8.
- Wilcox ML, Stewart JT. HPLC determination of guaiphenesin with selected medications on underivatized silica with an aqueous-organic mobile phase. J Pharm Biomed Anal 2000;23:909-16.
- McSharry WO, Savage IV. Simulteneous high-pressure liquid chromatographic determination of acetaminophen, guaiphenesin and dextromethorphan hydrochloride in cough syrup. J Pharm Sci 1980;69:212-4.
- Louhaichi NR, Jebali S, Loneslati MH, Adhoum N, Monsen L. Simultaneous determination of pseudoephedrine, pheniramine, guaiphenesin, pyrilamine, chlorpheniramine and dextromethorphan in cough and cold medicines by high performance liquid chromatography. Talanta 2009;78:991-7.
- Rauha JP, Salomies H, Aalto M. Simultaneous determination of bromhexine hydrochloride and methyl and propyl p-hydroxybenzoate and determination of dextromethorphan hydrochloride in cough-cold syrup by high-performance liquid chromatography. J Pharm Biomed Anal 1996;15:287-93.
- Hiedamann DR. Rapid, stability-indicating, high-pessure liquid chromatographic determination of theophylline, guaiphenesin and benzoic acid in liquid and solid pharmaceutical dosage forms. J Pharm Sci 1979;68:530-2.
- Galli V, Barbas C. High-performance liquid chromatographic analysis of dextromethorphan, guaiphenesin and benzoate acid in a cough syrup for stability testing. J Chromatogr A 2004;1048:207-11.
- Grosa G, Del Grosso E, Russo R, Allegrove G. Simultaneous, stability-indicating, HPLC-DAD determination of guaiphenesin and methyl and propyl-parabens in cough syrup. J Pharm Biomed Anal 2006;41:798-803.
- Daraghmeh N, Al-Omari MM, Sara Z, Badwan AA, Jaber AM. Determination of terbutalinesulphate and its degradation products in pharmaceutical formulations using LC. J Pharm Biomed Anal 2002;29:927-37.
- United States Pharmacopoeia/National Formulatory, 32nd ed. Rockville, MD: Pharmacopoeial Convention; 2009.
- British Pharmacopoeia, British Pharmacopoeial Commission Market Towers: London; 2010, Vol. 1, p.289.
- British Pharmacopoeia, British Pharmacopoeial Commission Market Towers: London; 2010, Vol. 2, p.2051.
- Ahuja S. Impurities evaluation of pharmaceuticals, Special Indian ed. New York: Marcel Dekker Inc. Publication; 2006. p. 96.
- ICH, Q3B (R2), Impurities in New Drug Substances, HarmonisedTripartite Guideline, 2006. p. 1-11.