- Corresponding Author:
- Ola I. A. Salem
Department of Pharmaceutical Organic Chemistry, Faculty of Pharmacy, Assiut University, Assiut-71526, Egypt. E−mail: olasalem69@gmail.com
| Date of Submission | 16 March 2013 |
| Date of Revision | 20 June 2013 |
| Date of Acceptance | 03 July 2013 |
| Indian J Pharm Sci 2013;75(5):545-556 |
Abstract
Two new series of 3-[2-(3,4-disubstituted-2,3-dihydrothiazol-2-ylidene)hydrazonopropylidenyl]-2-(methylthio)-3H-[1,2,4]triazolo[1,5-a]benzimidazole (6-29) and 3-[2-(3-substituted-4-oxothiazolidin-2-ylidene)hydrazonopropylidenyl]-2-(methylthio)-3H-[1,2,4]triazolo[1,5-a]benzimidazole (30-33) were synthesised starting from 1-[2-(methylthio)-3H-[1,2,4]triazolo[1,5-a]benzimidazol-3-yl] acetone N 4 -alkyl (aryl) thiosemicarbazones (2-5). Chemical structures of the compounds have been elucidated by different spectral data as well as elemental microanalysis. The newly synthesised compounds were tested for their in vitro antimicrobial activity using the standard agar cup diffusion method. Results revealed that most of the test compounds showed promising broad spectrum antibacterial and antifungal profiles against tested microorganisms, relative to references.
Keywords
[1,2,4]triazolo[1,5−a]benzimidazole, thiosemicarbazones, thiazolines, thiazolidinones, antimicrobial agents
There is a worldwide current interest in the development of new antimicrobial agents due to the increasing emergence of bacterial resistance to antibiotic therapy and to the newly emerging pathogens. In previous works, 2,3−disubstituted- 3H- [1,2,4]triazolo[1,5−a]benzimidazoles [1−3], showed equipotent antitubercular activity as that of isoniazide. Meanwhile, literature survey revealed that various thiosemicarbazone derivatives exhibited antimicrobial [4], antimalarial [5], antiamoebic [6], antitrypanosomal [7] and antitumour [8] effects. Likewise, Thiazolidinones showed antiinflammatory [9], antibacterial [10], antifungal [11], antitubercular [12], anticonvulsant [13] and anticancer [14] activities. In addition, other thiazolines exhibited antimicrobial [15], antiinflammatory [16], antitumour [17] and antioxidant [18]effects.
Development of new potent antimicrobial agents still a challenge and motivated by the abovementioned data, the present work aims at the synthesis and antimicrobial activity investigations, of two new series of 1−[2−(methylthio)−3H−[1,2,4]triazolo[1,5-a] benzimidazol-3-yl]acetone thiosemicarbazones (2−5) incorporating thiazolines (6−29), and thiazolidinones (30−33). It is hoped that combination of such rings in the new molecular design might lead to enhancement of the biological activities.
Materials and Methods
All compounds 1−33 were synthesised by following Scheme 1 and substituents are illustrated in Table 1. Melting points were determined in an electrothermal melting point apparatus (Stuart Scientific, Staffordshine, STIS, SMP3, UK) and were uncorrected. Follow up of the chemical reactions was carried out by TLC using precoated silica gel plates (kieselgel 0.25 mm, 60G F254, Merck, Germany) and developed by n-hexane:ethyl acetate as mobile phase. Visualization of the spots was effected by ultraviolet lamp (Spectroline, model cm−10, USA) at wavelengths 254 and 365 nm.
| Compd. no. | R1 | R2 |
|---|---|---|
| 2 | C6H5 | - |
| 3 | C2H5 | - |
| 4 | i−Pr | - |
| 5 | cyclo−C6H11 | - |
| 6 | C6H5 | H |
| 7 | C6H5 | Br |
| 8 | C6H5 | Cl |
| 9 | C6H5 | NO2 |
| 10 | C6H5 | OCH3 |
| 11 | C6H5 | CH3 |
| 12 | C2H5 | H |
| 13 | C2H5 | Br |
| 14 | C2H5 | Cl |
| 15 | C2H5 | NO2 |
| 16 | C2H5 | OCH3 |
| 17 | C2H5 | CH3 |
| 18 | i−Pr | H |
| 19 | i−Pr | Br |
| 20 | i−Pr | Cl |
| 21 | i−Pr | NO2 |
| 22 | i−Pr | OCH3 |
| 23 | i−Pr | CH3 |
| 24 | cyclo−C6H11 | H |
| 25 | cyclo−C6H11 | Br |
| 26 | cyclo−C6H11 | Cl |
| 27 | cyclo−C6H11 | NO2 |
| 28 | cyclo−C6H11 | OCH3 |
| 29 | cyclo−C6H11 | CH3 |
| 30 | C6H5 | |
| 31 | C2H5 | |
| 32 | i−Pr | |
| 33 | cyclo−C6H11 |
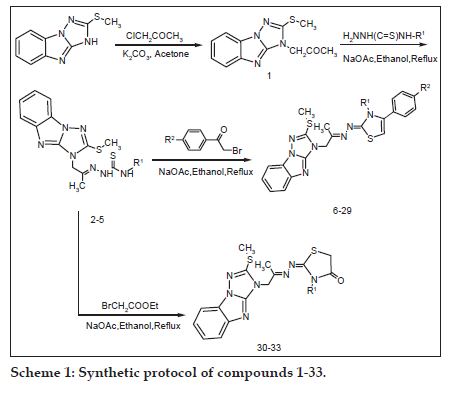
Scheme 1: Synthetic protocol of compounds 1−33.
IR spectra were carried out as KBr discs on a Shimadzu IR−470 Spectrometer (Shimadzu, Japan) at the Faculty of Pharmacy, Assiut University. 1H−NMR spectra were scanned on a Varian EM−360L NMR spectrometer (60 MHz, Varian, USA) at the Faculty of Pharmacy, Assiut University. 13C−NMR spectra were scanned on a Jeol JNM−LA series 400 FT NMR system (400 MHZ, Jeol, Japan) at the Assiut University, Unit of Trace Analysis. Chemical shifts are expressed in δ values (ppm) relative TMS as an internal standard; using CDCl3, unless otherwise specified, as a solvent, and D2O was used for the detection of the exchangeable protons.
Electron Impact mass spectra (EI−MS) were recorded on Gas Chromatography Mass Quadruple−2010 Plus mass spectrometer at 70 eV (Shimadzu, Japan) at the Unit of Microanalysis, Faculty of Science, Cairo University. Elemental microanalyses were performed on Vario EL III elemental analyzer (Vario, Germany) at the Unit of Microanalysis, Faculty of Science, Cairo University. The starting material 2−(methylthio)−3H−[1,2,4] triazolo[1,5−a]benzimidazole was prepared according to a reported procedure [2].
Synthesis of 1−[2−(methylthio)−3H−[1,2,4] triazolo[1,5−a]benzimidazol−3−yl] acetone (1):
The procedure for synthesis of 1−[2−(methylthio)−3H−[1,2,4]triazolo[1,5−a] benzimidazol−3−yl] acetone (1) is given as follows, to a suspension of 2−(methylthio)−3H−[1,2,4] triazolo[1,5−a]benzimidazole (1.02 g, 5 mmole) and potassium carbonate (0.70 g, 5 mmole) in dry acetone (15 ml), chloroacetone (0.4 ml, 5 mmole) was added. The reaction mixture was stirred for 15 h at ambient temperature. Acetone was evaporated and the residue was triturated with water then extracted with chloroform (3×15 ml). The chloroform extract was washed with water, dried over anhydrous magnesium sulphate and evaporated. The product was crystallised from dimethylformamide (DMF):water as colourless crystals.
Yield: 89%, mp: 230−232º, IR (cm−1): 3030 (Ar−H); 2890 (C−H aliphatic); 1715 (C=O); 1612, 1581, 1486 (C=C, C=N); 737 (Ar−H). 1H NMR (60 MHz): 2.20 (s, 3H, S−CH3), 2.60 (s, 3H, O=C−CH3), 4.80 (s, 2H, N−CH2), 6.80−7.95 (m, 4H, Ar−H). (Elemental analysis, calculated for C12H12N4OS Molecular weight (MW) 260.31): C, 55.37; H, 4.65; N, 21.52; Found: C, 55.20; H, 4.60; N, 21.40.
1−[2−(methylthio)−3H−[1,2,4]triazolo[1,5−a] benzimidazol−3−yl] acetone N4−alkyl (aryl) thiosemicarbazones (2−5)
To a solution of 1 (10 mmol) in absolute ethanol (20 ml), the appropriately substituted thiosemicarbazide[19] (10 mmol) was added. The reaction mixture was heated under reflux for 8−10 h, concentrated, cooled and diluted with water. The formed precipitate was filtered and recrystallised from absolute ethanol.
1−[2−(methylthio)−3H−[1,2,4]triazolo[1,5−a] benzimidazol-3−yl] acetone N4−phenyl thiosemicarbazone (2). Yield: 83%, mp: 205−207º, IR (cm−1): 3235, 3220 (N−H); 3045 (Ar−H); 2905 (C−H aliphatic); 1610, 1573, 1520, 1482 (C=C, C=N); 739 (Ar−H). 1H NMR (60 MHz, DMSO−d6): 2.00 (s, 3H, S−CH3), 2.50 (s, 3H, N=C−CH3), 5.05 (s, 2H, N=C−CH2), 6.95−7.90 (m, 9H, Ar−H), 9.00 (s, 1H, N−NH), 10.60 (br s, 1H, HN−C6H5). Anal. calcd. for C19H19N7S2 (MW=409.53): C, 55.72; H, 4.68; N, 23.94; Found: C, 55.53; H, 4.70; N, 24.06.
1−[2−(methylthio)−3H−[1,2,4]triazolo[1,5−a] benzimidazol−3−yl] acetone N4−ethyl thiosemicarbazone (3). Yield: 85%, mp: 187−189º, IR (cm−1): 3340, 3185 (N−H); 3055 (Ar−H); 2925 (C−H aliphatic); 1610, 1569, 1521, 1485 (C=C, C=N); 734 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.00 (t, 3H, N−CH2CH3), 1.90 (s, 3H, S−CH3), 2.65 (s, 3H, N=C−CH3), 3.55 (q, 2H, HN−CH2− CH3), 4.95 (s, 2H, N=C−CH2), 6.85 (t, 1H, HN−C2H5), 7.10−7.80 (m, 4H, Ar−H), 8.80 (s, 1H, N−NH). Anal. calcd. for C15H19N7S2 (MW=361.49): C, 49.84; H, 5.30; N, 27.12; Found: C, 49.66; H, 5.09; N, 27.19.
1−[2−(methylthio)−3H−[1,2,4]triazolo[1,5−a] benzimidazol−3−yl] acetone N4−isopropyl thiosemicarbazone (4). Yield: 82%, mp: 195−197º, IR (cm−1): 3310, 3160 (N−H); 3025 (Ar−H); 2900 (C−H aliphatic); 1607, 1570, 1512, 1487 (C=C, C=N); 740 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.00 (d, 6H, −CH(CH3)2), 1.90 (s, 3H, S−CH3), 2.65 (s, 3H, N=C−CH3), 4.00−4.70 (m, 1H, −CH(CH3)2), 4.90 (s, 2H, N=C−CH2), 6.50 (d, 1H, HN−C3H7), 7.10−7.80 (m, 4H, Ar−H), 8.55 (s, 1H, N−NH). 13C−NMR (400 MHz): 13.82 (S−CH3), 14.49 (N=C−CH3), 22.04 (-CH(CH3)2), 46.08 (−CH(CH3)2), 48.9 (N=C−CH2), 110.75, 110.95, 122.27, 123.75, 124.24, 134.05, 142.73, 154.43 (8 C, ArC’s), 165.64 (N=C), 176.38 (S=C). Anal. calcd. for C16H21N7S2 (MW=375.51): C, 51.18; H, 5.64; N, 26.11; Found: C, 51.06; H, 5.69; N, 26.25.
1−[2−(methylthio)−3H−[1,2,4]triazolo[1,5−a] benzimidazol−3−yl] acetone N4−cyclohexyl thiosemicarbazone (5). Yield: 78%, mp: 209−211º, IR (cm−1): 3320, 3130 (N−H); 3055 (Ar−H); 2915 (C−H aliphatic); 1612, 1585, 1520, 1507 (C=C, C=N); 738 (Ar−H). 1H NMR (60 MHz, CDCl3): 0.7−1.75 (m, 10H, (CH2)5 of cyclohexyl), 1.95 (s, 3H, S−CH3), 2.65 (s, 3H, N=C−CH3), 3.7− 4.3 (m, 1H, NCH of cyclohexyl), 4.90 (s, 2H, N=C−CH2), 6.70 (d, 1H, HN−C6H11), 7.10−7.80 (m, 4H, Ar−H), 8.70 (s, 1H, N−NH). Anal. calcd. for C19H25N7S2 (MW 415.58): C, 54.91; H, 6.06; N, 23.59; Found: C, 55.10; H, 5.90; N, 23.75.
3−[2−(3,4−disubstituted−2,3−dihydrothiazol−2−ylidene) hydrazonopropylidenyl]−2−(methylthio)−3H−[1,2,4] triazolo[1,5−a]benzimidazoles (6−29):
To a solution of the appropriately substituted thiosemicarbazone (2−5) (10 mmol) in absolute ethanol (20 ml), p-(un)substituted phenacyl bromide [20] (10 mmol) was added. The reaction mixture was heated under reflux for 12−24 h, partially concentrated and cooled. A saturated solution of sodium acetate (20 ml) was added. The formed precipitate was filtered and recrystallised from the appropriate solvent.
3−[2−(3,4−Diphenyl−2,3−dihydrothiazol−2−ylidene) hyd r a zonopropy l idenyl ] − 2 − (me thyl thio) − 3H−[1,2,4]triazolo[1,5−a]benzimidazole (6). Yield: 68%, mp: 131−133º (methanol), IR (cm−1): 3100 (Ar−H); 2900 (C−H aliphatic); 1613, 1578, 1523, 1480 (C=C, C=N); 731 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.80 (s, 3H, S−CH3), 2.60 (s, 3H, N=C−CH3), 4.85 (s, 2H, N=C−CH2), 5.95 (s, 1H, CH of thiazoline), 6.80−7.70 (m, 14H, Ar−H). EI−MS (m/z, % base): 509.45 (M+, 19.89), 70.05 (100). Anal. calcd. for C27H23N7S2 (MW=509.65): C, 63.63; H, 4.55; N, 19.24; Found: C, 63.90; H, 4.20; N, 19.33.
3 − [ 2 − ( 3 − P h e n y l − 4 − ( 4 − b r o m o p h e n y l ) − 2 , 3−dihydrothiazol−2−ylidene)hydrazonopropylidenyl]−2− (methylthio)−3H−[1,2,4]triazolo[1,5−a]benzimidazole (7). Yield: 71%, mp: 119−121º (ethanol), IR (cm−1): 3045 (Ar−H); 2905 (C−H aliphatic); 1613, 1568, 1531, 1465 (C=C, C=N); 824,734 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.80 (s, 3H, S−CH3), 2.65 (s, 3H, N=C−CH3), 5.00 (s, 2H, N=C−CH2), 6.10 (s, 1H, CH of thiazoline), 6.80−7.40 (m, 13H, Ar−H). Anal. calcd. for C27H22BrN7S2 (MW=588.55): C, 55.10; H, 3.77; N, 16.66; Found: C, 55.00; H, 4.00; N, 16.55.
3 − [ 2 − ( 3 − P h e n y l − 4 − ( 4 − c h l o r o p h e n y l ) − 2 , 3 − dihydrothiazol−2−ylidene)hydrazonopropylidenyl]−2− (methylthio)−3H−[1,2,4]triazolo[1,5−a]benzimidazole (8). Yield: 75%, mp: 108−110º (ethanol), IR (cm−1): 3075 (Ar−H); 2895 (C−H aliphatic); 1612, 1575, 1544, 1470 (C=C, C=N); 804,736 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.80 (s, 3H, S−CH3), 2.65 (s, 3H, N=C−CH3), 4.90 (s, 2H, N=C−CH2), 5.95 (s, 1H, CH of thiazoline), 6.90−7.60 (m, 13H, Ar−H). Anal. calcd. for C27H22ClN7S2 (MW=544.09): C, 59.60; H, 4.08; N, 18.02; Found: C, 59.50; H, 3.80; N, 17.85.
3−[2−(3−Phenyl−4−(4−nitrophenyl)−2,3− dihydrothiazol−2− ylidene)hydrazonopropylidenyl]−2− (methylthio)−3H− [1,2,4]triazolo[1,5−a]benzimidazole (9). Yield: 85%, mp: 115−117º (DMF/water), IR (cm−1): 3085 (Ar−H); 2950 (C−H aliphatic); 1607, 1561, 1523, 1477 (C=C, C=N); 1500, 1328 (NO2); 801, 845 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.85 (s, 3H, S−CH3), 2.65 (s, 3H, N=C−CH3), 4.90 (s, 2H, N=C−CH2), 5.95 (s, 1H, CH of thiazoline), 6.60−7.30 (m, 13H, Ar−H). Anal. calcd. for C27H22N8O2S2 (MW=554.64): C, 58.47; H, 4.00; N, 20.20; Found: C, 58.60; H, 3.79; N, 19.90.
3−[2−(3−Phenyl−4−(4−methoxyphenyl)−2,3− dihydrothiazol−2− ylidene)hydrazonopropylidenyl]−2− ( m e t h y l t h i o ) − 3 H − [ 1 , 2 , 4 ] t r i a z o l o [ 1 , 5 − a ] benzimidazole (10). Yield: 80%, mp: 207−209º (DMF/ water), IR (cm−1): 3030 (Ar−H); 2905 (C−H aliphatic); 1606, 1572, 1529, 1487 (C=C, C=N); 1200 (C−O); 836, 738 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.85 (s, 3H, S−CH3), 2.65 (s, 3H, N=C−CH3), 3.65 (s, 3H, O−CH3), 4.90 (s, 2H, N=C−CH2), 5.90 (s, 1H, CH of thiazoline), 6.60−7.20 (m, 13H, Ar−H). Anal. calcd. for C28H25N7OS2 (MW=539.67): C, 62.32; H, 4.67; N, 18.17; Found: C, 62.25; H, 4.40; N, 17.70.
3 − [ 2 − ( 3 − P h e n y l − 4 − ( 4 − m e t h y l p h e n y l ) − 2 , 3 − dihydrothiazol−2−ylidene)hydrazonopropylidenyl]−2− ( m e t h y l t h i o ) − 3 H − [ 1 , 2 , 4 ] t r i a z o l o [ 1 , 5 − a ] benzimidazole (11). Yield: 87%, mp: 123−125º (DMF/ water), IR (cm−1): 3080 (Ar−H); 2900 (C−H aliphatic); 1609, 1574, 1526, 1482 (C=C, C=N); 802, 737 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.85 (s, 3H, S−CH3), 2.25 (s, 3H, C6H4−CH3), 2.65 (s, 3H, N=C−CH3), 4.90 (s, 2H, N=C−CH2), 5.95 (s, 1H, CH of thiazoline), 6.80−7.30 (m, 13H, Ar−H). Anal. calcd. for C28H25N7S2 (MW=523.67): C, 64.22; H, 4.81; N, 18.72; Found: C, 64.70; H, 4.95; N, 18.81.
3−[2−(3−Ethyl−4−phenyl−2,3−dihydrothiazol−2−ylidene) hydrazonopropylidenyl]−2−(methylthio)−3H−[1,2,4] triazolo[1,5−a]benzimidazole (12). Yield: 79%, mp: 115−117º (ethanol), IR (cm−1): 3100 (Ar−H); 2920 (C−H aliphatic); 1612, 1572, 1531, 1480 (C=C, C=N); 740 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.25 (t, 3H, N−CH2CH3), 2.00 (s, 3H, S−CH3), 2.70 (s, 3H, N=C−CH3), 3.70 (q, 2H, NCH2−CH3), 4.95 (s, 2H, N=C−CH2), 5.75 (s, 1H, CH of thiazoline), 7.00−7.80 (m, 9H, Ar−H). Anal. calcd. for C23H23N7S2 (MW=461.61): C, 59.84; H, 5.02; N, 21.24; Found: C, 59.80; H, 5.40; N, 21.17.
3−[2−(3−Ethyl−4−(4−bromophenyl)−2, 3−dihydrothiazol−2− ylidene)hydrazonopropylidenyl]−2−(methylthio)− 3H−[1,2,4]triazolo[1,5−a]benzimidazole (13). Yield: 85%, mp: 155−157º (DMF/water), IR (cm−1): 3020 (Ar−H); 2950 (C−H aliphatic); 1614, 1572, 1527, 1479 (C=C, C=N); 808, 730 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.15 (t, 3H, N−CH2CH3), 1.95 (s, 3H, S−CH3), 2.65 (s, 3H, N=C−CH3), 3.65 (q, 2H, NCH2−CH3), 4.85 (s, 2H, N=C−CH2), 5.70 (s, 1H, CH of thiazoline), 6.90−7.60 (m, 8H, Ar−H). Anal. calcd. for C23H22BrN7S2 (MW=540.50): C, 51.11; H, 4.10; N, 18.14; Found: C, 50.95; H, 3.93; N, 18.01.
3−[2−(3−Ethyl−4−(4−chlorophenyl)−2,3−dihydrothiazol−2− ylidene)hydrazonopropylidenyl]−2−(methylthio)− 3H− [1,2,4]triazolo[1,5−a]benzimidazole (14). Yield: 80%, mp: 163−165º (hexane/ethyl acetate), IR (cm−1): 3050 (Ar−H); 2955 (C−H aliphatic); 1614, 1572, 1529, 1480 (C=C, C=N); 809, 730 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.15 (t, 3H, N−CH2CH3), 2.00 (s, 3H, S−CH3), 2.70 (s, 3H, N=C−CH3), 3.70 (q, 2H, NCH2−CH3), 4.95 (s, 2H, N=C−CH2), 5.75 (s, 1H, CH of thiazoline), 7.00−7.90 (m, 8H, Ar−H). Anal. calcd. for C23H22ClN7S2 (MW=496.05): C, 55.69; H, 4.47; N, 19.77; Found: C, 55.48; H, 4.46; N, 19.60.
3−[2−(3−Ethyl−4−(4−nitrophenyl)−2,3−dihydrothiazol−2− ylidene)hydrazonopropylidenyl]−2−(methylthio)− 3H−[1,2,4]triazolo[1,5−a]benzimidazole (15). Yield: 82%, mp: 174−176º (hexane/ethyl acetate), IR (cm−1): 3070 (Ar−H); 2950 (C−H aliphatic); 1614, 1567, 1537, 1458 (C=C, C=N); 1504, 1333(NO2); 846, 731 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.15 (t, 3H, N−CH2CH3), 2.00 (s, 3H, S−CH3), 2.70 (s, 3H, N=C−CH3), 3.75 (q, 2H, NCH2−CH3), 4.95 (s, 2H, N=C−CH2), 5.90 (s, 1H, CH of thiazoline), 7.00−8.30 (m, 8H, Ar−H). Anal. calcd. for C23H22N8O2S2 (MW=506.60): C, 54.53; H, 4.38; N, 22.12; Found: C, 54.10; H, 4.09; N, 21.98.
3 − [ 2 − ( 3 − E t h y l − 4 − ( 4 − m e t h o x y p h e n y l ) − 2 , 3 − dihydrothiazol−2− ylidene)hydrazonopropylidenyl]−2− ( m e t h y l t h i o ) − 3 H − [ 1 , 2 , 4 ] t r i a z o l o [ 1 , 5 − a ] benzimidazole (16). Yield: 77%, mp: 170−172º (ethanol), IR (cm−1): 3080 (Ar−H); 2895 (C−H aliphatic); 1610, 1577, 1531, 1487 (C=C, C=N); 1220 (C−O); 813, 731 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.15 (t, 3H, N−CH2CH3), 2.00 (s, 3H, S−CH3), 2.65 (s, 3H, N=C−CH3), 3.70 (q, 2H, NCH2−CH3), 3.80 (s, 3H, O−CH3), 4.95 (s, 2H, N=C−CH2), 5.75 (s, 1H, CH of thiazoline), 6.70−7.75 (m, 8H, Ar−H). Anal. calcd for C24H25N7OS2 (MW=491.63): C, 58.63; H, 5.13; N, 19.94; Found: C, 58.53; H, 5.14; N, 19.96.
3 − [ 2 − ( 3 − E t h y l − 4 − ( 4 − m e t h y l p h e n y l ) − 2 , 3 − dihydrothiazol−2−ylidene)hydrazonopropylidenyl]−2− ( m e t h y l t h i o ) − 3 H − [ 1 , 2 , 4 ] t r i a z o l o [ 1 , 5 − a ] benzimidazole (17). Yield: 84%, mp: 160−162º (dioxane/ether), IR (cm−1): 3080 (Ar−H); 2880 (C−H aliphatic); 1613, 1575, 1529, 1486 (C=C, C=N); 804, 730 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.10 (t, 3H, N−CH2CH3), 1.95 (s, 3H, S−CH3), 2.30 (s, 3H, C6H4−CH3), 2.65 (s, 3H, N=C−CH3), 3.65 (q, 2H, N−CH2−CH3), 4.90 (s, 2H, N=C−CH2), 5.65 (s, 1H, CH of thiazoline), 6.90−7.70 (m, 8H, Ar−H). Anal. calcd for C24H25N7S2 (MW=475.63): C, 60.60; H, 5.30; N, 20.61; Found: C, 60.30; H, 5.10; N, 20.86.
3-[2-(3-Isopropyl-4-phenyl-2,3-dihydrothiazol-2- ylidene)hydrazonopropylidenyl]-2-(methylthio)- 3H-[1,2,4]triazolo[1,5-a]benzimidazole (18). Yield: 73%, mp: 172−174º (ethanol), IR (cm−1): 3095 (Ar−H); 2950 (C−H aliphatic); 1612, 1576, 1532, 1479 (C=C, C=N); 736, 693 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.40 (d, 6H, −CH(CH3)2), 1.95 (s, 3H, S−CH3), 2.65 (s, 3H, N=C−CH3), 3.70−4.25 (m, 1H, −CH(CH3)2), 4.90 (s, 2H, N=C−CH2), 5.65 (s, 1H, CH of thiazoline), 7.00−7.70 (m, 9H, Ar−H). Anal. calcd for C24H25N7S2 (MW=475.63): C, 60.60; H, 5.30; N, 20.61; Found: C, 60.45; H, 5.29; N, 20.53.
3-[2-(3-Isopropyl-4-(4-bromophenyl)-2,3- dihydrothiazol-2-ylidene)hydrazonopropylidenyl]- 2- ( m e t h y l t h i o ) -3H- [ 1 , 2 , 4 ] t r i a z o l o [ 1 , 5 - a ] benzimidazole (19). Yield: 76%, mp: 190−192º (ethanol), IR (cm−1): 3045 (Ar−H); 2905 (C−H aliphatic); 1613, 1568, 1531, 1465 (C=C, C=N); 824, 734 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.45 (d, 6H, −CH(CH3)2), 2.00 (s, 3H, S−CH3), 2.65 (s, 3H, N=C−CH3), 3.70−4.25 (m, 1H, −CH(CH3)2), 4.95 (s, 2H, N=C−CH2), 5.75 (s, 1H, CH of thiazoline), 7.00−8.10 (m, 8H, Ar−H). EI−MS (m/z, % base): 556.30 (M+2, 29.21), 555.30 (M+1, 94.73), 554.30 (M+, 28.34), 553.30 (M+−l, 89.54), 204.10 (100). Anal. calcd for C24H24BrN7S2 (MW=554.53): C, 51.98; H, 4.36; N, 17.68; Found: C, 50.95; H, 4.20; N, 17.50.
3-[2-(3-Isopropyl-4-(4-chlorophenyl)-2,3- dihydrothiazol-2-ylidene)hydrazonopropylidenyl]- 2- ( m e t h y l t h i o ) -3H- [ 1 , 2 , 4 ] t r i a z o l o [ 1 , 5 - a ] benzimidazole (20). Yield: 80%, mp: 176−178º (ethanol), IR (cm−1): 3015 (Ar−H); 2900 (C−H aliphatic); 1618, 1573, 1538, 1470 (C=C, C=N); 804, 736 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.40 (d, 6H, −CH(CH3)2), 1.95 (s, 3H, S−CH3), 2.65 (s, 3H, N=C−CH3), 3.70−4.25 (m, 1H, −CH(CH3)2), 4.95 (s, 2H, N=C−CH2), 5.65 (s, 1H, CH of thiazoline), 7.00−7.80 (m, 8H, Ar−H). 13C−NMR (400 MHz): 14.69 (S−CH3), 15.33 (N=C−CH3), 18.88 (−CH(CH3)2), 50.23 (−CH(CH3)2), 50.79 (N=C−CH2), 100.09 (C5 of thiazoline), 100.60, 110.39, 112.03, 121.71, 123.48, 124.39, 128.96, 130.15, 130.67, 134.34, 135.10, 139.77, 152.29, 154.61 (15C, ArC’s), 165.29 (H3C−C=N), 168.67 (C2 of thiazoline). Anal. calcd for C24H24ClN7S2 (MW=510.07): C, 56.51; H, 4.74; N, 19.22; Found: C, 56.71; H, 4.80; N, 19.08.
3−[2−(3−Isopropyl−4−(4−nitrophenyl)−2,3− dihydrothiazol −2−ylidene)hydrazonopropylidenyl]−2−(methylthi o)−3H−[1,2,4]triazolo[1,5−a]benzimidazole (21). Yield: 85%, mp: 162−164º (DMF/water), IR (cm−1): 3085 (Ar−H); 2960 (C−H aliphatic); 1607, 1574, 1536, 1461 (C=C, C=N); 1505, 1334 (NO2); 848, 741 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.40 (d, 6H, −CH(CH3)2), 1.95 (s, 3H, S−CH3), 2.65 (s, 3H, N=C−CH3), 3.70−4.25 (m, 1H, −CH(CH3)2), 4.90 (s, 2H, N=C−CH2), 5.75 (s, 1H, CH of thiazoline), 7.00−8.30 (m, 8H, Ar−H). Anal. calcd for C24H24N8O2S2 (MW=520.63): C, 55.37; H, 4.65; N, 21.52; Found: C, 55.28; H, 4.80; N, 21.67.
3−[2−(3−Isopropyl−4−(4−methoxyphenyl)−2,3− dihydrothiazol−2−ylidene)hydrazonopropylidenyl]−2− ( m e t h y l t h i o ) − 3 H − [ 1 , 2 , 4 ] t r i a z o l o [ 1 , 5 − a ] benzimidazole (22). Yield: 81%, mp: 167−169º (hexane/ethyl acetate), IR (cm−1): 3080 (Ar−H); 2990 (C−H aliphatic); 1610, 1576, 1533, 1480 (C=C, C=N); 1200 (C−O); 809, 734 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.40 (d, 6H, −CH(CH3)2), 1.95 (s, 3H, S−CH3), 2.65 (s, 3H, N=C−CH3), 3.75 (s, 3H, O−CH3), 3.60−4.25 (m, 1H, −CH(CH3)2), 4.90 (s, 2H, −N=C−CH2), 5.60 (s, 1H, CH of thiazoline), 6.50−8.00 (m, 8H, Ar−H). Anal. calcd for C25H27N7OS2 (MW=505.66): C, 59.38; H, 5.38; N, 19.39; Found: C, 59.28; H, 5.32; N, 19.06.
3−[ 2 − (3−Isopropyl−4−(4−methylphenyl ) − 2 ,3− dihydrothiazol−2−ylidene)hydrazonopropylidenyl]−2− (methylthio)−3H−[1,2,4]triazolo[1,5−a]benzimidazole (23). Yield: 88%, mp: 178−180º (ethanol), IR (cm−1): 3050 (Ar−H); 2900 (C−H aliphatic); 1612, 1578, 1531, 1486 (C=C, C=N); 808, 736 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.50 (d, 6H, −CH(CH3)2), 2.05 (s, 3H, S−CH3), 2.40 (s, 3H, C6H4−CH3), 2.75 (s, 3H, N=C−CH3), 3.80−4.30 (m, 1H, −CH(CH3)2), 5.05 (s, 2H, N=C−CH2), 5.75 (s, 1H, CH of thiazoline), 7.20−7.70 (m, 8H, Ar−H). Anal. calcd for C25H27N7S2 (MW=489.66): C, 61.32; H, 5.56; N, 20.02; Found: C, 61.13; H, 5.51; N, 19.89.
3−[2−(3−Cyclohexyl−4−phenyl−2,3−dihydrothiazol−2− ylidene)hydrazonopropylidenyl]−2−(methylthio)−3H− [1,2,4]triazolo[1,5−a]benzimidazole (24). Yield: 74%, mp: 150−152º (DMF/water), IR (cm−1): 3050 (Ar−H); 2985 (C−H aliphatic); 1612, 1578, 1529, 1481 (C=C, C=N); 732 (Ar−H). 1H NMR (60 MHz, CDCl3): 0.80−1.80 (m, 10H, (CH2)5 of cyclohexyl), 2.00 (s, 3H, S−CH3), 2.70 (s, 3H, N=C−CH3), 3.00−4.00 (m, 1H, NCH of cyclohexyl), 5.00 (s, 2H, N=C−CH2), 5.75 (s, 1H, CH of thiazoline), 7.10−7.80 (m, 9H, Ar−H). Anal. calcd for C27H29N7S2 (MW=515.69): C, 62.88; H, 5.67; N, 19.01; Found: C, 62.75; H, 5.55; N, 19.10.
3-[2-(3-Cyclohexyl-4-(4-bromophenyl)-2,3- dihydrothiazol-2- ylidene) hydrazonopropylidenyl]- 2- ( m e t h y l t h i o ) -3H- [ 1 , 2 , 4 ] t r i a z o l o [ 1 , 5 - a ] benzimidazole (25). Yield: 84%, mp: 98−100º (hexane/ethyl acetate), IR (cm−1): 3040 (Ar−H); 2925 (C−H aliphatic); 1619, 1575, 1534, 1479 (C=C, C=N); 840, 736 (Ar−H). 1H NMR (60 MHz, CDCl3): 0.80−1.90 (m, 10H, (CH2)5 of cyclohexyl), 2.00 (s, 3H, S−CH3), 2.65 (s, 3H, N=C−CH3), 3.00−3.90 (m, 1H, NCH of cyclohexyl), 4.90 (s, 2H, N=C−CH2), 5.65 (s, 1H, CH of thiazoline), 6.90−8.00 (m, 8H, Ar−H). EI−MS (m/z, % base): 596.45 (M++2, 23.49), 594.45 (M+, 24.00), 55.10 (100). Anal. calcd for C27H28BrN7S2 (MW=594.59): C, 54.54; H, 4.75; N, 16.49; Found: C, 54.40; H, 4.65; N, 16.40.
3−[2−(3−Cyclohexyl−4−(4−chlorophenyl)−2,3− dihydrothiazol−2−ylidene)hydrazonopropylidenyl]−2−(m ethylthio)−3H−[1,2,4]triazolo[1,5−a]benzimidazole (26). Yield: 80%, mp: 104−106º (ethanol), IR (cm−1): 3100 (Ar−H); 2900 (C−H aliphatic); 1617, 1575, 1532, 1479 (C=C, C=N); 829, 735 (Ar−H). 1H NMR (60 MHz, CDCl3): 0.80−1.75 (m, 10H, (CH2)5 of cyclohexyl), 2.00 (s, 3H, S−CH3), 2.65 (s, 3H, N=C−CH3), 3.00−3.60 (m, 1H, NCH of cyclohexyl), 4.90 (s, 2H, −N=C−CH2), 5.85 (s, 1H, CH of thiazoline), 7.00−7.60 (m, 8H, Ar−H). Anal. calcd for C27H28ClN7S2 (MW=550.14): C, 58.95; H, 5.13; N, 17.82; Found: C, 58.80; H, 4.90; N, 17.65.
3-[2-(3-Cyclohexyl-4-(4-nitrophenyl)-2,3- dihydrothiazol-2-ylidene)hydrazonopropylidenyl]- 2- ( m e t h y l t h i o ) -3H- [ 1 , 2 , 4 ] t r i a z o l o [ 1 , 5 - a ] benzimidazole (27). Yield: 83%, mp: 102−104º (DMF/ water), IR (cm−1): 3100 (Ar−H); 2995 (C−H aliphatic); 1616, 1571, 1536, 1459 (C=C, C=N); 1504, 1331 (NO2); 843, 736 (Ar−H). 1H NMR (60 MHz, CDCl3): 0.80−1.75 (m, 10H, (CH2)5 of cyclohexyl), 2.00 (s, 3H, S−CH3), 2.65 (s, 3H, N=C−CH3), 3.00−3.70 (m, 1H, NCH of cyclohexyl), 4.90 (s, 2H, N=C−CH2), 5.75 (s, 1H, CH of thiazoline), 7.10−7.70 (m, 8H, Ar−H). Anal. calcd for C27H28N8O2S2 (MW=560.69): C, 57.84; H, 5.03; N, 19.98; Found: C, 57.85; H, 5.21; N, 19.65.
3-[2-(3-Cyclohexyl-4-(4-methoxyphenyl)-2,3- dihydrothiazol-2-ylidene)hydrazonopropylidenyl]- 2-(methylthio)-3H-[1,2,4]triazolo[1,5-a] benzimidazole (28). Yield: 82%, mp: 162−164º (hexane/ethyl acetate), IR (cm−1): 3100 (Ar−H); 2990 (C−H aliphatic); 1610, 1576, 1533, 1480 (C=C, C=N); 1200 (C−O); 808, 734 (Ar−H). 1H NMR (60 MHz, CDCl3): 0.80−1.80 (m, 10H, (CH2)5 of cyclohexyl), 2.00 (s, 3H, S−CH3), 2.65 (s, 3H, N=C−CH3), 3.20−3.70 (m, 1H, NCH of cyclohexyl), 3.80 (s, 3H, O−CH3), 5.00 (s, 2H, N=C−CH2), 5.75 (s, 1H, CH of thiazoline), 6.70−7.70 (m, 8H, Ar−H). Anal. calcd for C28H31N7OS2 (MW=545.72): C, 61.62; H, 5.73; N, 17.97; Found: C, 61.70; H, 5.56; N, 17.70.
3-[2-(3-Cyclohexyl-4-(4-methylphenyl)-2,3- dihydrothiazol-2-ylidene) hydrazonopropylidenyl]-2- (methylthio)-3H-[1,2,4]triazolo[1,5-a]benzimidazole (29). Yield: 78%, mp: 188−190º (DMF/water), IR (cm−1): 3065 (Ar−H); 2915 (C−H aliphatic); 1612, 1581, 1520, 1488 (C=C, C=N); 804, 737 (Ar−H). 1H NMR (60 MHz, CDCl3): 0.80−1.80 (m, 10H, (CH2)5 of cyclohexyl), 1.95 (s, 3H, S−CH3), 2.35 (s, 3H, C6H4−CH3), 2.65 (s, 3H, N=C−CH3), 3.20−3.80 (m, 1H, NCH of cyclohexyl), 4.90 (s, 2H, N=C−CH2), 5.65 (s,1H, CH of thiazoline), 7.00−7.80 (m, 8H, Ar−H). Anal. calcd for C28H31N7S2 (MW=529.72): C, 63.49; H, 5.90; N, 18.51; Found: C, 63.28; H, 5.96; N, 18.75.
3−[2−(3−substituted−4−oxothiazolidin−2−ylidene)hydraz onopropylidenyl]−2−(methylthio)−3H− [1,2,4] triazolo [1,5−a]benzimidazole (30−33)
Ethyl bromoacetate (1.67 g, 1.1 ml, 0.01 mol) was added to a solution of the appropriately substituted thiosemicarbazones (2−5) (0.01 mol) in absolute ethanol (20 ml). The reaction mixture was heated under reflux for 9−12 h. After cooling, a saturated solution of sodium acetate (20 ml) was added. The precipitated product was filtered and recrystallised from the appropriate solvent.
3-[2-(3-Phenyl-4-oxothiazolidin-2-ylidene) hydrazonopropylidenyl]-2-(methylthio)-3H-[1,2,4] triazolo[1,5-a]benzimidazole (30). Yield: 79%, mp: 219−221º (ethanol), IR (cm−1): 3050 (Ar−H); 2920 (C−H aliphatic); 1716 (C=O); 1620, 1565, 1485 (C=C, C=N); 735 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.90 (s, 3H, S−CH3), 2.70 (s, 3H, N=C−CH3), 3.85 (s, 2H, CH2 of thiazolidinone), 4.95 (s, 2H, N=C−CH2), 7.00−7.80 (m, 9H, Ar−H). Anal. calcd for C21H19N7OS2 (MW=449.55): C, 56.11; H, 4.26; N, 21.81; Found: C, 56.30; H, 4.15; N, 21.50.
3-[2-(3-Ethyl-4-oxoth iazo l i d in-2- y l idene) hydrazonopropylidenyl]-2-(methylthio)-3H-[1,2,4] triazolo[1,5-a]benzimidazole (31). Yield: 81%, mp: 194−196º (ethanol), IR (cm−1): 3040 (Ar−H); 2895 (C−H aliphatic); 1699 (C=O); 1635, 1500, 1487 (C=C, C=N); 727 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.20 (t, 3H, N−CH2CH3), 2.00 (s, 3H, S−CH3), 2.65 (s, 3H, N=C−CH3), 3.55 (s, 2H, CH2 of thiazolidinone), 3.70 (q, 2H, N−CH2−CH3), 4.90 (s, 2H, N=C−CH2), 7.00−7.70 (m, 4H, Ar−H). EI−MS (m/z, % base): 403.40 (M+2, 12.40), 402.40 (M+1, 23.52), 401.40 (M+, 100). Anal. calcd for C17H19N7OS2 (MW=401.51): C, 50.85; H, 4.77; N, 24.42; Found: C, 50.40; H, 4.43; N, 24.40.
3−[2−(3−Isopropyl−4−oxothiazolidin−2−ylidene)hy drazonopropylidenyl]−2−(methylthio)−3H−[1,2,4] triazolo[1,5−a]benzimidazole (32). Yield: 80%, mp: 186−188º (hexane/ethyl acetate), IR (cm−1): 3050 (Ar−H); 2970 (C−H aliphatic); 1705 (C=O); 1616, 1552, 1480 (C=C, C=N); 740 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.40 (d, 6H, −CH(CH3)2), 2.00 (s, 3H, S−CH3), 2.65 (s, 3H, N=C−CH3), 3.55 (s, 2H, CH2 of thiazolidinone), 4.40−4.70 (m, 1H, −CH(CH3)2), 4.95 (s, 2H, N=C−CH2), 7.00−7.70 (m, 4H, Ar−H). 13C− NMR (400 MHz): 14.65 (S−CH3), 15.59 (N=C−CH3), 18.72 (CH(CH3)2), 32.34 (CH2 of thiazolidinone), 48.07 (−CH(CH3)2), 49.52 (N=C−CH2), 110.57, 111.46, 121.97, 123.55, 124.45, 134.26, 154.60, 159.85 (8 C, ArC’s), 163.42 (C2 of thiazolidinone), 165.39 (H3C−C=N), 171.92 (C=O). EI−MS (m/z, % base): 417.40 (M+2, 4.08), 416.40 (M+1, 7.63), 415.40 (M+, 33.33), 80.00 (100). Anal. calcd for C18H21N7OS2 (MW=415.53): C, 52.03; H, 5.09; N, 23.60; Found: C, 51.90; H, 5.20; N, 23.50.
3-[2-(3-Cyclohexyl-4-oxothiazolidin-2-ylidene) hydrazonopropylidenyl]-2-(methylthio)-3H-[1,2,4] triazolo[1,5-a]benzimidazole (33). Yield: 82%, mp: 218−220º (hexane/ethyl acetate), IR (cm−1): 3050 (Ar−H); 2905 (C−H aliphatic); 1705 (C=O); 1617, 1535, 1481 (C=C, C=N); 741 (Ar−H). 1H NMR (60 MHz, CDCl3): 1.00−1.85 (m, 10H, (CH2)5 of cyclohexyl), 2.00 (s, 3H, S−CH3), 2.65 (s, 3H, N=C−CH3), 3.55 (s, 2H, CH2 of thiazolidinone), 4.00−4.70 (m, 1H, NCH of cyclohexyl), 4.95 (s, 2H, N=C−CH2), 7.10−7.80 (m, 4H, Ar−H). Anal. calcd for C21H25N7OS2 (MW=455.60): C, 55.36; H, 5.53; N, 21.52; Found: C, 55.28; H, 5.38; N, 21.67.
Antimicrobial activity
All newly synthesised compounds were tested for their in vitro antibacterial and antifungal activities, in comparison to ampicillin (for antibacterial investigation), fluconazole and clotrimazole (for antifungal investigation) as reference drugs, respectively, (at a concentration of 100 mmol/ml), using the standard agar cup diffusion method[21]. The assay was done at Assiut University Mycological Center (AUMC), Faculty of Science, Assiut University. Six bacterial species representing both Gram−positive and Gram−negative strains were selected and they are common contaminants of the environment in Egypt and some of which are involved in human and animal diseases. The used Gram−negative bacterial strains are Serratia marcescens (AUMC B55), Pseudomonas aeruginosa (AUMC B73), and Escherichia coli (AUMC B53), while the Gram−positive strains were represented by Staphylococcus aureus (AUMC B54), Bacillus cereus (AUMC B52), and Micrococcus luteus (AUMC B112). Three human pathogenic fungi species [Trichophyton rubrum AUMC 1804, Candida albicans AUMC 1299 and Scopulariopsis brevicaulis AUMC 361], a phytopathogenic fungi (Fusarium oxysporum AUMC 5119) and food deteriorating fungal species (Aspergillus flavus AUMC 1276 and Geotrichum candidum AUMC 226) were used in the present study. For antibacterial investigation; bacterial strains were individually cultured for 48 h in 100 ml conical flasks containing 30 ml nutrient agar (NA) medium. While, in antifungal testing; spore suspension in sterile distilled water was prepared from 7 days old culture of the test fungi growing on Sabouraud’s dextrose broth (30 ml) media in 100 ml conical flasks. The final spore concentration was nearly 5×104 spores/ml. Bioassay was done in 10 cm sterile dishes in which 1 ml of bacterial or spore suspension and 15 ml of NA were poured. Plates were shaken gently to homogenise the inocula. After solidification of the media, 5 mm cavities were cut in the solidified agar (4 cavities/plate) using sterile cork borer. The test compounds (1−33) and reference drugs were dissolved in dimethyl sulphoxide (100 mmol/ml) and were loaded in the cavities. In addition, other cavities were loaded with the solvent, dimethyl sulphoxide (DMSO) and served as a negative control. The seeded plates were incubated at 28±2° for 48 h for bacteria and for 7 days in case of fungi. The radii of inhibition zones (in mm) of triplicate sets were measured and the results are cited in Tables 2 and 3, respectively for bacteria and fungi.
| Compd. no. | Zone of inhibition in mm | |||||
|---|---|---|---|---|---|---|
| Gram−positive | Gram−negative | |||||
| S. aureus | B. cereus | M. luteus | E. coli | P. aeruginosa | S. marcescens | |
| (AUMCB54)a | (AUMCB52) | (AUMCB112) | (AUMCB73) | (AUMCB55) | ||
| DMSOb | − | − | − | − | − | − |
| 1 | 10 (6.25) | 10 (12.5) | 12 (12.5) | 11 (0.78) | 16 (100) | 12 (12.5) |
| 2 | 12 (25) | 12 (50) | 12 (100) | 12 (50) | 12 (100) | 10 (6.25) |
| 3 | 10 (1.56) | 10 (25) | 14 (12.5) | 14 (0.78) | 12 (6.25) | 12 (0.39) |
| 4 | 10 (100) | − | − | 13 (100) | 10 (100) | 14 (100) |
| 6 | 10 (6.25) | 15 (100) | 12 (50) | 10 (3.12) | 10 (25) | 10 (50) |
| 7 | 16 (100) | − | − | 15 (100) | 17 (100) | − |
| 9 | 10 (3.12) | 12 (25) | 14 (50) | 14 (12.5) | 11 (3.12) | 10 (1.56) |
| 10 | 12 (100) | − | − | 14 (100) | 14 (100) | 12 (100) |
| 12 | 11 (100) | − | − | 11 (100) | 12 (100) | − |
| 14 | 12 (12.5) | 10 (12.5) | − | 12 (25) | 10 (50) | 12 (100) |
| 15 | 10 (100) | 10 (100) | − | 11 (25) | 13 (100) | 10 (25) |
| 17 | − | − | − | 10 (100) | − | − |
| 18 | 17 (100) | 10 (6.25) | 13 (3.12) | 12 (3.12) | 12 (25) | 12 (1.56) |
| 19 | 12 (100) | − | − | 16 (100) | − | 12 (100) |
| 20 | 10 (6.25) | 12 (100) | − | 12 (1.56) | − | 10 (3.12) |
| 21 | 10 (100) | − | − | 15 (100) | 15 (100) | 13 (100) |
| 23 | 12 (12.5) | 10 (12.5) | 13 (50) | 12 (6.25) | 10 (25) | 10 (6.25) |
| 24 | 12 (100) | − | − | 10 (25) | 16 (100) | 15 (100) |
| 25 | 12 (100) | 12 (100) | − | 12 (25) | 14 (100) | 10 (6.25) |
| 26 | 14 (100) | 12 (50) | 12 (6.25) | 12 (25) | 13 (12.5) | 12 (0.78) |
| 27 | 10 (100) | − | − | 14 (100) | − | 10 (12.5) |
| 29 | 14 (100) | 12 (100) | 12 (100) | 12 (12.5) | 18 (100) | 15 (6.25) |
| 30 | 12 (25) | 13 (100) | − | 10 (1.56) | 20 (100) | 15 (6.25) |
| 31 | 10 (100) | − | − | 15 (100) | 13 (100) | 18 (100) |
| 32 | 13 (25) | 10 (25) | 13 (100) | 12 (25) | 13 (100) | 10 (6.25) |
| 33 | 18 (100) | 16 (100) | 12 (12.5) | 12 (25) | 15 (6.25) | 12 (0.78) |
| Ampicillin | 12 (50) | 13 (50) | 13 (25) | 12 (25) | 12 (6.25) | 10 (0.78) |
The values in the paranthesis are MIC in μmol/ml; aAUMC: Assiut University Mycological Center, bDMSO: Dimethyl sulphoxide, (-): Resistant
Table 2: In vitro antibacterial activity of the test active compounds and ampicillin
| Compd. no. | Zone of inhibition in mm | |||||
|---|---|---|---|---|---|---|
| C. albicans | S. brevicaulis | T. rubrum | A. flavus | F. oxysporum | G. candidum | |
| (AUMC1299)a | (AUMC361) | (AUMC1804) | (AUMC1276) | (AUMC5119) | (AUMC226) | |
| DMSOb | − | − | − | − | − | − |
| 2 | 12 (100) | 16 (6.25) | 12 (0.1) | 12 (0.2) | 12 (3.125) | 18 (0.78) |
| 5 | − | − | − | − | − | 8 (100) |
| 6 | 12 (100 ) | 10 (6.25) | 14 (0.1 ) | 15 (0.39 ) | 13( 6.25) | 18 (1.56 ) |
| 7 | − | 10 (12.5) | 18 (0.39 ) | 12 (0.39) | 10 (100) | 16 (1.56) |
| 11 | − | 10 (12.5) | 24 (0.2) | 12 (0.2) | − | 18( 0.78) |
| 15 | 9 (100) | 10 (25) | − | − | 10 (100) | − |
| 16 | − | 15 (50) | 14 (0.05) | 14 (0.39) | 16 (100) | 15 (0.39) |
| 22 | 22p.i (100) | 14 (25) | 18 (0.1) | 14 (0.2) | 12 (6.25) | 18 (0.78) |
| 29 | 10 (100) | 14 (6.25) | 16 (0.1) | 14 (0.39) | 10 (0.39) | 19 (0.78) |
| 30 | 12 (100) | 14 (6.25) | 18 (0.1) | 13 (0.39) | 14 (0.78) | 15 (0.2) |
| Fluconazole | 20 p.i (25) | − | 10 (100) | 10 (12.5) | − | 16 (1.56) |
| Clotrimazole | Not tested | 24p.i (1.56) | Not tested | Not tested | 15 (0.39) | Not tested |
The values in the paranthesis are MIC in μmol/ml; aAUMC: Assiut University Mycological Center, bDMSO: Dimethyl sulphoxide, (-): Resistant, p.i.: partial inhibition
Table 3: In vitro antifungal activity of the test active compounds, FLUCONAZOLE and CLOTRIMAZOLE
Results and Discussion
Synthetic procedures adopted to obtain the target compounds are outlined in Scheme 1. The starting compound 2−(methylthio)−3H−[1,2,4] triazolo[1,5−a]benzimidazole was prepared according to a reported procedure and its structure was confirmed by matching its physical and spectral data with the reported one [2]. The key intermediate 1−[2−(methylthio)−3H−[1,2,4] triazolo[1,5−a]benzimidazol−3−yl]acetone (1) was prepared by reaction of chloroacetone with 2−(methylthio)−3H−[1,2,4]triazolo[1,5−a]benzimidazole and potassium carbonate in dry acetone. IR spectrum of intermediate 1 showed band due to carbonyl group at 1715 cm−1 and disappearance of NH band at 3300 cm−1. Its 1H−NMR spectrum showed appearance of two new singlets at d 4.8 and 2.2 ppm attributed to the methylene and methyl groups, respectively with the disappearance of NH proton.
The new intermediate thiosemicarbazone derivatives (2−5), were prepared by refluxing compound 1 with the appropriate 4−substituted−3- thiosemicarbazide[19] in absolute ethanol. IR spectra of the thiosemicarbazones (2−5) showed NH band at 3130−3340 cm−1 and disappearance of C=O band at 1715 cm−1. Their 1H−NMR spectra proved two singlets (D2O−exchangeable) at ranges of d 6.50−10.60 and 8.55-9.00 ppm indicating two NH protons. Furthermore, 13C−NMR spectrum of compound 4 displayed two essential signals at d 176.38 ppm and d 165.64 ppm corresponding to C=S and C=N of the thiosemicarbazone moiety.
In addition, treatment of thiosemicarbazone derivatives (2−5) with p−(un)substituted phenacyl bromides[20], in the presence of anhydrous sodium acetate, afforded the corresponding thiazoline derivatives (6−29). Structure elucidation of compounds (6−29) was established by IR spectra which showed disappearance of the two NH protons of thiosemicarbazones. 1H−NMR spectra of these compounds were characterised by the disappearance of the two NH protons of thiosemicarbazone derivatives and appearance of a singlet at a range of d 5.60−6.10 ppm corresponding to thiazoline=CH proton. 13C−NMR spectrum of compound 20 showed disappearance of the thione signal and appearance of a signal at d 168.67 ppm attributable to C=N at C2 of thiazoline ring which reveals the cyclization of the thiosemicarbazone, and formation of thiazoline ring. The EI−mass spectra of compounds 6, 18 and 25 showed their molecular ion peaks (M+) at m/z 509, 554 and 594, respectively. In addition, compounds 18 and 25 revealed prominent isotope peaks (M+2) at m/z 556 and 596, respectively reflecting 81Br. Peaks at m/z 553, 555 and m/z 593, 595 in mass spectra of compounds 18 and 25, respectively, were assigned to (M+−H) fragment and their isotopes.
In continue, synthesis of compounds (30−33), were established through heating thiosemicarbazones (2−5) with ethyl bromoacetate in the presence of anhydrous sodium acetate. IR spectra showed the disappearance of NH bands of the thiosemicarbazone moiety and the appearance of a new band at a range of 1699−1716 cm−1 attributed to a carbonyl group of thiazolidinone. 1H−NMR spectra lacked signals characteristic of the two NH protons and showed a new singlet at a range of d 3.55−3.85 ppm attributed to methylene C5 protons, beside the disappearance of the thione group peak. 13C−NMR spectrum of compound 32 revealed the appearance of two new signals at d 32.34 and 171.92 ppm which are attributed to the methylene and amidic carbonyl group of thiazolidin−4−one ring, respectively. The EI−MS of compounds 31 and 32 showed molecular ion peaks at m/z 401 and 415, respectively.
The antibacterial activities for all new compounds (1−33) were compared with ampicillin at 100 μmol/ml using standard agar cup diffusion method [21]. Dimethyl sulphoxide (DMSO) showed no antimicrobial activity against tested strains. In general, Gram−negative bacterial strains were susceptible for inhibition by tested compounds more than Gram−positive ones. Analysis of the results (Table 2) revealed that the key intermediate 1 showed higher activity than ampicillin by multiple folds, against S. aureus (8 folds), B. cereus (4 folds), M. luteus (2 folds) and E. coli (32 folds). On the other hand, thiosemicarbazones (2−5), showed that all compounds were more or less active except compound 5 (R1=cyclo−C6H11). The most active thiosemicarbazone was compound 3 (R1=C2H5) which showed a broad spectrum inhibition for all tested Gram−positive and Gram−negative bacterial strains. MICs for 3 ranged from (0.39−25 μmol/ml), that means it has 1−32 folds increasing activity than ampicillin, against all tested bacterial strains. The above results indicate that the presence of bulky groups (R1=i−Pr or C6H5) may decrease or abolish (R1=cyclo−C6H11) activity in thiosemicarbazones.
The most active compounds in thiazoline series (R1=C6H5) were compounds 6 and 9 (R2=H, NO2, respectively). Compound 6 showed higher activity than ampicillin by about 8 folds against S. aureus and E. coli. Compound 9 exhibited double−fold activities than ampicillin against B. cereus, E. coli and P. aeruginosa while 16−fold increase in activity relative to ampicillin against S. aureus. Conclusion of these data indicates that presence of nitro group increases the activity while, presence of chloro or methyl groups abolishes it.
Substitution at para position with chloro and nitro groups in thiazoline derivatives 14 and 15 (R1=C2H5, R2=Cl, NO2, respectively) demonstrated good activity against tested bacterial strains. Both compounds were equipotent with ampicillin against E. coli. Furthermore, compound 14 showed 4 fold increasing in activity relative to ampicillin against S. aureus and B. cereus.
In addition, cyclization of thiosemicarbazone 3 (R1=i−Pr) to the corresponding thiazolines, e, g. compound 18 (R2=H) surpasses ampicillin activity by 8 fold against B. cereus, M. luteus and E. coli. Also, compound 20 (R2=Cl) has 8 fold higher activity than reference drug against S. aureus and 16 fold against E. coli. Compound 23 (R2=CH3) exhibited 4 folds increasing in activity against S. aureus, B. cereus and E. coli relative to ampicillin.
Thiazoline derivatives 24, 25 and 26 (R1=cyclo−C6H11, R2=H, Br, Cl, respectively) were equipotent to ampicillin against E. coli. In addition, compound 26 (R2=Cl) showed 4 fold higher activity against M. Luteus and compound 29 (R2=CH3) elicited double fold activity against E. coli. The most active thiazolidinone derivatives were compounds 30, 32 and 33. Compound 30 (R1=C6H5) showed double fold activity than ampicillin against S. aureus. and 16 folds against E. coli. Compound 32 (R1=i−Pr) showed double fold increase in activity than ampicillin against S. aureus and B. cereus. Compound 33 (R1=cyclo−C6H11) exhibited double fold activity than ampicillin against M. luteus while it was equipotent with ampicillin against all tested Gram−negative bacteria. Most compounds are inactive against M. luteus except compounds 1, 2, 3, 6, 9, 18, 23, 26, 29, 32 and 33. MICs of active compounds and ampicillin against tested Gram−positive and Gram−negative bacteria were shown in Table 2.
Antifungal activities were assayed for all new compounds (1−33) using standard agar cup diffusion method[21] relative to fluconazole and clotrimazole as references (at 100 μmol/ml). Results of the antifungal activity, (Table 3) revealed that only ten compounds showed strong activities against the used fungal strains. They were active against S. brevicaulis except thiosemicarbazone 5 (R1=cyclo−C6H11) and active against F. oxysporum except compounds 5 and 11. It is noteworthy to mention that, fluconazole was completely inactive against F. oxysporum and S. brevicaulis. Among thiosemicarbazone derivatives only compounds 2 and 5 (R1=C6H5, cyclo−C6H11, respectively) were active. Compound 2 showed good activity compared to fluconazole.
Cyclization of thiosemicarbazone compound 2 (R1=C6H5) to the corresponding thiazoline derivatives (6−11) led to three active compounds 6, 7 and 11 (R2=H, Br, CH3, respectively). The active compounds showed antifungal activity against S. brevicaulis and F. oxysporum. Compound 6 elicited higher activity than fluconazole against T. rubrum (1000 folds) and A. flavus (32 folds). Compound 7 exhibited higher activity than fluconazole against T. rubrum (256 folds) and A. flavus (32 folds). Compound 11 proved higher activity than fluconazole against G. candidum (2 folds), T. rubrum (500 folds) and A. flavus (64 folds). Compounds 7 and 11 (R2=Br, CH3, respectively) were inactive against C. albicans. In addition, compound 11 (R2=CH3) was inactive against F. oxysporum.
From the ethyl series 3 (R1=C2H5), only two compounds (15 and 16, R2=NO2, OCH3, respectively) were active. The most potent compound was compound 16 (R2=OCH3) (4−2000 folds). It was more potent than fluconazole against G. candidum (4 folds), T. rubrum (2000 folds) and A. flavus (32 folds) but was inactive against C. albicans.
Converting thiosemicarbazone compound 4 (R1=i−Pr) to derivatives (18−23) led to only one active compound 22 (R2=OCH3). It was more potent than fluconazole against G. candidum (2 folds), T. rubrum (1000 folds) and A. flavus (64 folds).
Thiazoline derivatives (24−29) derived from thiosemicarbazone compound, 5 (R1=cyclo−C6H11) showed no activity except compound 29 (R2=CH3), it showed higher activity against all fluconazole resistant strains. It was more potent than fluconazole against G. candidum (2 folds), T. rubrum (1000 folds) and A. flavus (32 folds) while, it was equipotent with clotrimazole against F. oxysporum (MIC=0.39 μmol/ml).
Antifungal activity of thiazolidinone derivatives (30−33), revealed that only compound 30 (R1=C6H5) was active against all strains. It was more potent than fluconazole against G. candidum (8 folds), T. rubrum (1000 folds) and A. flavus (32 folds). MICs of active compounds and references against tested fungal species are listed in Table 3.
In conclusion, the objective of this study was to synthesise and investigate the antimicrobial activities of some new 2,3−disubstituted−3H−[1,2,4]triazolo[1,5−a] benzimidazole derivatives incorporating thiazoline and thiazolidinone rings. The obtained results clearly revealed that most of these derivatives showed promising broad spectrum antibacterial and antifungal activities compared to ampicillin, fluconazole and clotrimazole, respectively.
References
- Mohamed BG, Abdel−Alim AA, Hussein MA. Synthesis of 1−acyl−2−alkylthio−1,2,4−triazolobenzimidazoles with antifungal, antiinfammatory and analgesic effects. Acta Pharm 2006;56:31.
- Mohamed BG, Hussein MA, Abdel−Alim AA, Hashem M. Synthesis and antimicrobial activity of some new 1−alkyl−2−alkylthio−1,2,4−triazolobenzimidazole derivatives. Arch Pharm Res 2006;29:26−33.
- Mohammed AF, Hussein MA, Abdel−Moty SG, Abdel−Alim AA. Synthesis and biological evaluation of some benzimidazo−1,2,4−triazole derivatives as antimicrobial and antiinfammatory agents. Bull Pharm SciAssiutUniv 2011;34:77−92.
- Omar A, Ahmed I, Hassan A, AboulWafa O, Abou−Shleib H, Ismail K. Synthesis and evaluation for antibacterial and antifungal activities of new 1−phenylhydrazono−2−(substituted thiocarbamoyl) hydrazonopyruvaldehyde and the corresponding thiazoline and thiazolidinone derivatives. Alex J Pharm Sci 1990;4:182−6.
- Scovill JP, Klayman DL, Lambros C, Childs GE, Notsch JD. 2−Acetylpyridine thiosemicarbazones. 9. Derivatives of 2−acetylpyridine 1−oxide as potential antimalarial agents. J Med Chem 1984;27:87−91.
- Sharma S, Athar F, Maurya MR, Naqvi F, Azam A. Novel bidentate complexes of Cu (II) derived from 5−nitrofuran−2−carboxaldehyde thiosemicarbazones with antiamoebic activity against E. histolytica. Eur J Med Chem 2005;40:557−62.
- Fujii N, Mallari JP, Hansell EJ, Mackey Z, Doyle P, Zhou YM, et al. Discovery of potent thiosemicarbazone inhibitors of rhodesain and cruzain. Bioorg Med ChemLett 2005;15:121−3.
- Perez−Rebolledo A, Ayala JD, de Lima GM, Marchini N, Bombieri G, Zani CL, et al. Structural studies and cytotoxic activity of N (4)−phenyl−2−benzoylpyridine thiosemicarbazoneSn (IV) complexes. Eur J Med Chem 2005;40:467−72.
- Bekhit AA, Fahmy HT, Rostom SA, El−Din ABekhit A. Synthesis and biological evaluation of some thiazolylpyrazole derivatives as dual antiinflammatory antimicrobial agents. Eur J Med Chem 2010;45:6027−38.
- Sayyed M, Mokle S, Bokhare M, Mankar A, Surwase S, Bhusare S, et al. Synthesis of some new 2,3−diaryl−1,3−thiazolidin−4−ones asantibacterial agents. Arkivoc 2006;2:187−92.
- Ramachandran R, Rani M, Kabilan S. Design, synthesis and biological evaluation of novel 2−[(2,4−diaryl−3−azabicyclo[3.3.1]nonan−9−ylidene) hydrazono]−1,3−thiazolidin−4−ones as a new class of antimicrobial agents. Bioorg Med ChemLett 2009;19:2819−23.
- Küçükgüzel ŞG, Oruç EE, Rollas S, Şahin F, Özbek A. Synthesis, characterisation and biological activity of novel 4−thiazolidinones, 1,3,4−oxadiazoles and some related compounds. Eur J Med Chem 2002;37:197−206.
- Shiradkar MR, Ghodake M, Bothara KG, Bhandari SV, Nikalje A, Akula KC, et al. Synthesis and anticonvulsant activity of clubbed thiazolidinone–barbituric acid and thiazolidinone–triazole derivatives. Arkivoc 2007;14:58−74.
- Havrylyuk D, Mosula L, Zimenkovsky B, Vasylenko O, Gzella A, Lesyk R. Synthesis and anticancer activity evaluation of 4−thiazolidinones containing benzothiazole moiety. Eur J Med Chem 2010;45:5012−21.
- Bondock S, Khalifa W, Fadda AA. Synthesis and antimicrobial evaluation of some new thiazole, thiazolidinone and thiazoline derivatives starting from 1−chloro−3,4−dihydronaphthalene−2−carboxaldehyde. Eur J Med Chem 2007;42:948−54.
- Sondhi SM, Singh N, Lahoti AM, Bajaj K, Kumar A, Lozach O, et al. Synthesis of acridinyl−thiazolino derivatives and their evaluation for anti−infammatory, analgesic and kinase inhibition activities. Bioorg Med Chem 2005;13:4291−9.
- Mahler G, Serra G, Dematteis S, Saldaña J, Domínguez L, Manta E. Synthesis and biological evaluation of simplified mycothiazole analogues. Bioorg Med Chem 2006;16:1309−11.
- Shih MH, Ke FY. Syntheses and evaluation of antioxidant activity of sydnonyl substituted thiazolidinone and thiazoline derivatives. Bioorg Med Chem 2004;12:4633−43.
- Barton D, Ollis WD. Comprehensive Organic Chemistry. Vol. 3. New York: Pergamon Press; 1979. p. 453.
- Cowper RM, Davidson LH. Organic Syntheses. Vol. 2. New York: John Wiley and Sons; 1943. p. 480.
- William H. An introduction to quantitative principles and evaluation. New York: Academic Press; 1977, p. 17-68.