Huan Gao, Xingjun Wu, Lihua Chen, Liwen Zhang, Yingfang Zhang, Chenyang Zhang and Min Ye*
Department of Neurology, Affiliated BenQ Hospital of Nanjing Medical University, Nanjing 210019, China
- *Corresponding Author:
- Min Ye
Department of Neurology, Affiliated BenQ Hospital of Nanjing Medical University, Nanjing 210019, China
E-mail: zhuixian163661@163.com
| This article was originally published in a special issue, “Biomedical Research in Healthcare Setting” |
| Indian J Pharm Sci 2020:82(3)Spl issue7;39-47 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
To study the role of Neural precursor cell expressed developmentally down-regulated protein 4.1 on ubiquitination of central Insulin-like growth factor 1β pathway in Alzheimer’s Disease, and to clarify the molecular mechanism of Neural precursor cell expressed developmentally down-regulated protein 4.1 and Insulin-like growth factor 1β for central insulin signalling pathway damage. Mouse hippocampal neurons cells were isolated from Intracerebral haemorrhage mice. 30 μM or 300 μM S-Nitroso-l-cysteine were incubated with hippocampal neurons cells for 4 h. pCMV-Neural precursor cell expressed developmentally down-regulated protein 4.1 or siR- Neural precursor cell expressed developmentally down-regulated protein 4.1 were synthesized and transfected into S-Nitroso-l-cysteine treated hippocampal neurons cells. Cycloheximide and MG132 were incubated with hippocampal neuron cells after S-Nitroso-l-cysteine stimulation. Insulin resistant brain state model were constructed by streptozotocin injection. Western blot were used to test the expression of neural precursor cell expressed developmentally down-regulated protein 4.1, Insulin-like growth factor-1Rβ, Protein kinase B and p- Protein kinase B, Glycogen synthase kinase 3β and p- Glycogen synthase kinase 3β, Tau protein and p-Tau protein in vitro and in vivo. Coimmunoprecipitation were used to analyse Neural precursor cell expressed developmentally downregulated protein 4.1 and Insulin-like growth factor-1Rβ. Neural precursor cell expressed developmentally down-regulated protein 4.1 in the hippocampus of Tg2576 mice increased significantly compared to the control mice (p<0.05), while the expression of Insulin-like growth factor-1Rβ decreased significantly compared to the control mice (p<0.05). S-Nitroso-l-cysteine stimulated hippocampal neuron cells to increase the ubiquitination of Insulin-like growth factor-1Rβ (p<0.05) and decrease the expression of Insulin-like growth factor-1Rβ (p<0.05). Down-regulation of Neural precursor cell expressed developmentally downregulated protein 4.1 can decreased the ubiquitination of Insulin-like growth factor-1Rβ significantly (p<0.05), and rescued the S-Nitroso-l-cysteine inhibition effect on expression of Insulin-like growth factor-1Rβ. Over-expression Neural precursor cell expressed developmentally down-regulated protein 4.1 could increase the ubiquitination level of Insulin-like growth factor-1Rβ (p<0.05). MG132 and cyclohexide both can reverse the degradation of Insulin-like growth factor-1Rβ induced by S-Nitroso-l-cysteine. The phosphorylation degree of Protein kinase B and Glycogen synthase kinase 3β in neurons cells treated with S-Nitroso-l-cysteine decreased significantly (p<0.05), while Tau phosphorylation level in hippocampal neuron cells treated with Insulin-like growth factor-1 increased significantly (p<0.05). S-Nitroso-lcysteine can improve Tau protein phosphorylation (p<0.05) in neurons cells. C1060 site is necessary for its ubiquitination and degradation. Insulin-like growth factor-1Rβ were significantly lower (p<0.05) in the Streptozotocin model while Neural precursor cell expressed developmentally down-regulated protein 4.1 and Aβ were significantly higher (p<0.05) in the Streptozotocin model. p- Protein kinase B/Protein kinase B and p- Glycogen synthase kinase 3β/Glycogen synthase kinase 3β in the Streptozotocin dementia rats were significantly lower (p<0.05), and p-Tau/Tau were significantly increased (p<0.05). Abnormal increasing neural precursor cell expressed developmentally down-regulated protein 4.1 in brain injury tissue induced the rising ubiquitination of Insulin-like growth factor-1Rβ and rising of Aβ amount and Tau protein phosphorylation. C1060 site in Insulin-like growth factor-1Rβ was necessary for the Neural precursor cell expressed developmentally down-regulated protein 4.1 of ubiquitination and degradation of Insulin-like growth factor-1Rβ. In addition, Neural precursor cell expressed developmentally down- regulated protein 4.1 affects Insulin-like growth factor-1 signal pathway in brain, such as inhibiting Protein kinase B and Glycogen synthase kinase 3β phosphorylation, promoting Tau protein phosphorylation, and promoting β-amyloid secretions.
The Role of Neural Precursor Cell Expressed Developmentally Down-Regulated Protein 4.1- Mediated Insulin-Like Growth Factor-1Rβ Ubiquitination in the Pathogenesis of Alzheimer’s disease
Keywords
NEDD4.1, IGF-1Rβ, ubiquitination, Alzheimer’s disease
Alzheimer’s disease (AD) is a progressive neurodegenerative disease with main symptoms as memory loss, disorientation and cognitive ability recession [1]. AD patients need long-time and special nursing. With the increasing incidence rate of AD in China, the family and society suffer from heavy burden. The pathogenesis of AD is debatable now; there are diverse theory such as cholinergic nerve damage [2], β-amyloid protein and tau protein theory [3], inflammatory mechanism, oxidative stress and mitochondrial dysfunction [4], insulin resistance to explain the pathogenesis of AD [5]. There is no specific and efficient therapy and drug against AD, so pathogenesis and new drug of AD are one of the hottest topics in basic and clinical study. Epidemiological evidence showed that type II diabetes mellitus (2- Diabetes mellitus), whose main pathogenesis of insulin resistance, was one of the highest risk factors for AD patients [6]. The incidence rate of AD in type 2 diabetes patients is 2-3 times higher than diabetes-free people [7]. Insulin-like growth factor (IGF-1) is a neurotropic peptide with similar structure and function to insulin. Neurons can also synthesize a small amount of insulin and IGF-1 in brain [8].
It has recently discovered that Neural Precursor cell expressed development down regulated gene 4 (NEDD4) is a ubiquitin ligase E3 including two subclasses: NEDD4.1 and NEDD4. 2. The research on the specific substrate of NEDD4.1 in the central nervous system and its role attract researchers’ attention [9]. In our study, we focused on the study of protein post-translational modification of ubiquitination degradation pathway of central IGF-1 pathway, using in vitro cytology test and in vivo animal model to further clarify the molecular mechanism of central insulin signalling pathway damage, and further reveal the role of NEDD4.1 in the pathological mechanism of AD, in order to provide clues for mechanism research of AD, as well as for the study of therapeutic targets and drugs of AD.
Materials and Methods
Animals:
Intracerebral haemorrhage (ICH) mice, Sprague Dawley (SD) rats (200-250 g) and Tg2576 mice were purchased from Nanjing Junke Bioengineering Co., Ltd.
Main reagents and instruments:
Normal Donkey Serum (NDS), fetal bovine serum (a31608), trypsin (25 200-072) were purchased from GIBCO, USA; Dulbecco’s Modified Eagle Medium (DMEM) and neurobasic medium (BYX1564P) were purchased from BYXBIO, USA;MG132 (47 4790) and polyvinylidene fluoride (PVDF) membrane (IPFL00010) were purchased from Merck millipore, USA; Lipofectamine 2000TM were purchased from Invitrogen, USA. Recombinant IGF-1, 100×protease inhibitor, S-Nitroso-l-cysteine (SNOC), McAb, B27 and β-actin McAb were purchased from SIGMA Company; Radioimmunoprecipitation assay (RIPA) cell lysate (P0013D), bicinchoninic acid assay (BCA) (P0012S), enhanced luminol-based chemiluminescent (ECL) (P0018S) and BCA detection kit (P0010S) were purchased from biyuntian Biotechnology Research Institute; Rabbit anti IGF-1Rβ, NEDD4.1, protein kinase B (Akt), Glycogen synthase kinase-3β (GSK3β), Tau protein, p-Akt, p-GSK3β and p-Tau polyclonal antibodies and rabbit antiβ-actin polyclonal antibodies (AP0063) were purchased from BioWorld company, USA; Goat anti rabbit IgG (ZB-2301) was purchased from Beijing zhongshanjinqiao Biotechnology Co., Ltd. Frozen microtome from Leica (CM1850).
Isolation and culture of mouse hippocampal neurons cells:
The hippocampal tissues of ICH mice were isolated and chopped, then digested by 0.25 % trypsin Ethylenediaminetetraacetic acid (EDTA) at 37° for 20 min, and the hippocampal neurons were collected by centrifugation at 1000 rpm for 5 min. The cell suspension was obtained and diluted with DMEM medium containing 10 % fetal bovine serum (FBS). The cells were cultured in neurobasic medium containing 2 % B27 and 0.5 mm at 37°, and 10 μmol cytarabine was added to the medium for 2-3 d. The morphology of hippocampal neurons was observed by Atomic Force Microscope (AFM).
Over-expression and down-regulation of NEDD4.1 in hippocampal neurons cells:
pCMV-NEDD4.1 expression vector for overexpressing NEDD4.1 was purchased from Origin Company. siR-NEDD4.1 was synthesized by Shanghai Jikai Gene Chemistry Technology Co., Ltd. To synthesize siR-NEDD4.1, the forward primer was 5’-CCUACAGUAGUAGUAGUTT-3’ and reverse primer was 5’- AUUCAGUAGUAGGTT-3’. The forward primer of the negative control (NC) of siRNA was 5’-UUCUCCGAACGUGUCACGUT-3’, and the reverse primer was 5’-ACGUCUCGGAGAATT-3’. pCMV-NEDD4.1 and siR-NEDD4.1 was respectively transfected into mouse hippocampal neurons cells by Lipofectamine 2000TM.
Treatment with SNOC:
The neurons cells were cultured in neurobasic medium containing 2 % B27 for 48 h, and were grouped into three groups. SNOC NC group was incubated in neurobasic medium without SNOC, low SNOC group was incubated in neurobasic medium with 30 μM SNOC and high SNOC group was incubated with 300 μM SNOC solution at 37° for 4 h. SNOC is an efficient nitric oxide donor. The cells were collected for Immunocoprecipitation and Western blot analysis
Treatment of hippocampal neuron cells with Cycloheximide (CHX):
Mouse hippocampal neuron cells treated with SNOC previously or not were maintained in fresh DMEM culture in well. To inhibit protein synthesis, cells were treated with pro-apoptoticagent CHX (5 g/ml) for 0, 3, 6 and 9 h. All incubations were performed at 37° in a 5 % CO2 -95 % air mixture for variable periods of time. After treatment with CHX, all medium was discarded and cells were harvested for following experiments. Uninfected and untreated macrophages were used as control.
Treatment of hippocampal neuron cells with MG132:
To inhibit proteasome, the mouse hippocampal neuron cells treated with SNOC previously or not were incubated with or without 10 mM MG132 for 2 h at 37°, and cultured in Neurobasal medium under a humidified 5 % CO2 -95 % air mixtures at 37°. After treatment with MG132, all medium was discarded and cells were harvested for following Western Blotting to analyze IGF-1Rβ.
Stimulation of hippocampal neuron cells with IGF-1:
Hippocampal neuron cells treated with SNOC previously or not were maintained in DMEM supplemented with 10 % fetal bovine serum at 37° in a humidified 5 % CO2 -95 % air mixture. To determine the effect of IGF- 1 on AKT phosphorylation, hippocampal neuron cells were grown in 100-mm dishes to 90 % con-fluence and then incubated in DMEM without serum for 16 h before exposure to treatments. Recombinant IGF-1 was added to cell culture medium at a final concentration of 10 nM for 5 min. After treatment with IGF-1, all medium was discarded and cells were harvested for following Western Blotting to analyze AKT and p-AKT, GSK3β and p-GSK3β, Tau protein and p-Tau protein.
Animal model construction:
SD rats were fed on a high fat, high sugar and high protein diet for 2 mo. Streptozotocin (STZ) was dissolved in artificial cerebrospinal fluid (120 mM NaCl, 3 mM KCl, 1.15 mM CaCl2, 0.8 mM MgCl2, 0.33 mm NaH2PO4, 27 mm NaHCO3) and injected into the lateral ventricle of each SD rat at a dose of 1.25 mg/ kg to establish dementia model of insulin resistance. The parameters of bilateral intraventricular injection were: 0.1mm behind the anterior fontanelle, 2.5mm under the dura, and ±0.8mm side away; the parameters of bilateral intraventricular injection were: 0.8mm behind the anterior fontanelle, 4.0mm under the dura, and ±1.5mm side away. The injection time of each animal was 5 min and the needle was kept for 10 min. Active nitrogen injury model was taken as negative control group: each SD rat was injected with SIN-150 Ug (SIN-1 dissolved in normal saline) into the lateral ventricle.
After injection, the rats were selected by water maze and dark avoidance experiment. Rats were anesthetized with pentobarbital sodium (50 mg/kg). The aorta was perfused with 37° normal saline, and then perfused with 4 % paraformaldehyde at 4° for 30 min. At the end of perfusion, the brain tissue was taken out and dehydrated in 10 %, 20 % and 30 % sucrose solution successively at 4° until the tissue mass sank. The brain tissue was detected by Western blot and immunohistochemistry.
Coimmunoprecipitation (Co-IP):
The cells for analysis were lysed with RIPA solution, centrifuged at 12 000 r/min for 5 min to collect the supernatant. The supernatant was added into phenylmethylsulfonyl fluoride (PMSF) and mouse monoclonal anti hemagglutinin (HA) antibody, incubated at 4° for 8 h, then purified with Protein G-Agarose column, incubated for 2 h. The sample was centrifuged at 12 000 r/min for 5 min to remove the supernatant ,and the precipitation was washed by agarose beads with IP buffer for 5-6 times. Then sample buffer was add into the beads and mixed gently, and seal the column. The mixture was heated at 95° for 5min, and filter paper was used to absorb the water around the mixture. The mixture was added into the new micro centrifuge tube and was centrifuged at 12 00g for 30 s to obtain the immunoprecipitate.
Western Blot:
Western Blot (WB) was used to detect the expression of NEDD4.1 and IGF-IRβ in the hippocampus of Tg2576 mice, active nitrogen injury model and STZ dementia model, the lysate of hippocampal neurons cells and their immunoprecipitates after SNOC treatment in section “2.3.3” to “2.3.6”. The total protein was extracted and analyzed the protein content by Bicinchoninic Acid (BCA) kit. Polyacrylamide gel (4 % concentration gel, 10 % separation gel) electrophoresis at constant current was used to transfer the proteins to PVDF membrane. PVDF membrane was blocked by 5 % TBST milk powder solution for 1.5 h. primary antibody of IGF- 1Rβ, NEDD4.1, AKT, GSK3β, Tau protein, p-AKT, p-GSK3β and p-Tau protein were diluted to 1:1000- 1:4000 and incubated with PVDF membrane at 4° overnight. After rinsing by Phosphate-buffered saline (PBS) solution, the PVDF membrane was incubated with different IgG-HRPs (1:1000-1:4000) for 2-3h at 37°. ECL agent was added. The protein bands were scanned by gel scanner, and β-actin was used as internal reference.
Statistical methods:
SPSS 22.0 statistical software was used for data analysis. Data were present as (x±s). Student’s T-test was used for comparison between the two groups, and one-way ANOVA was used for comparison between multiple groups, p<0. 05 means there is statistical significance between different groups.
Results and Discussion
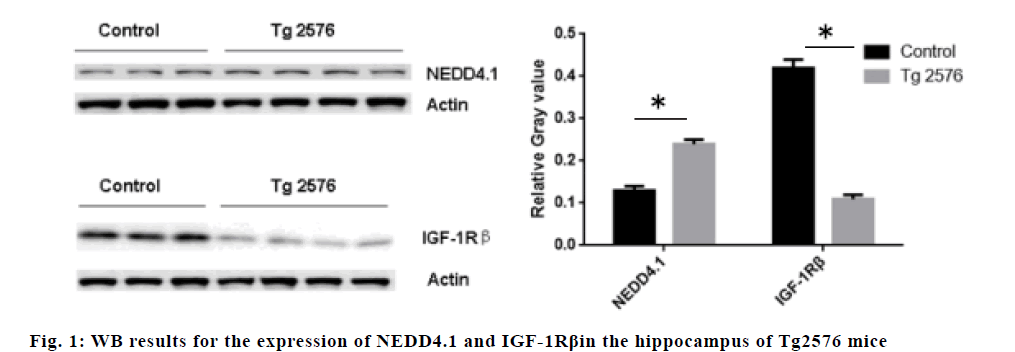
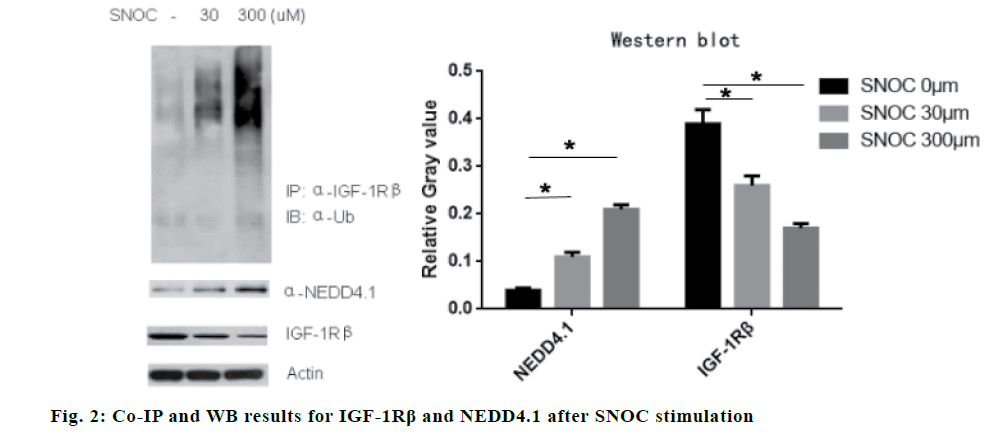
The expression of NEDD4.1 in the hippocampus of Tg2576 mice increased significantly compared to the control mice (p<0.05), while the expression of IGF- 1Rβ decreased significantly compared to the control mice (p<0.05), suggesting that NEDD4.1 may be involved in the IGF-1Rβ expression in vivo. The results are shown in fig. 1. The results showed that SNOC stimulated hippocampal neuron cells to increase the ubiquitination of IGF-1Rβ (p<0.05) and decrease the expression of IGF-1Rβ (p<0.05). The Co-IP experiment showed that SNOC enhanced the expression of NEDD4.1 (P<0.05). The results are shown in fig. 2.
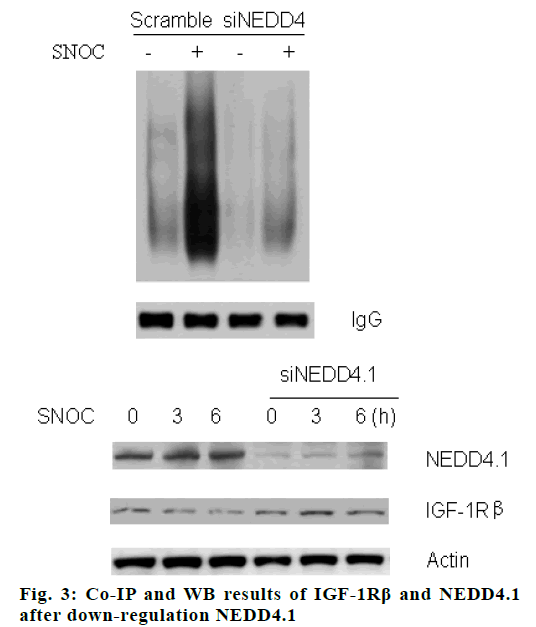
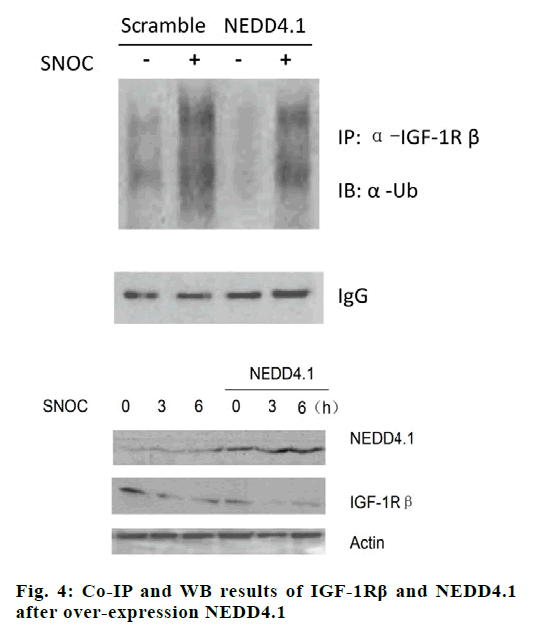
When NEDD4.1 gene was silenced by specific siRNA, the ubiquitination of IGF-1Rβ decreased significantly (p<0.05), and the down-regulation of NEDD4.1 rescued the SNOC inhibition effect on expression of IGF-1Rβ, the results are shown in fig. 3. After over-expression NEDD4.1 gene, the ubiquitination level of IGF-1Rβ increased (p<0.05), and the expression of IGF-1Rβ was inhibited (p<0.05), as shown in fig. 4.
Western blot test showed that the IGF-1Rβ in hippocampal neuron cells decreased significantly (p<0.05) after cyclohexide treatment, no matter after SNOC treatment or not and the decrease level was positively associated with the duration of CHX treatment, as shown in fig. 5.
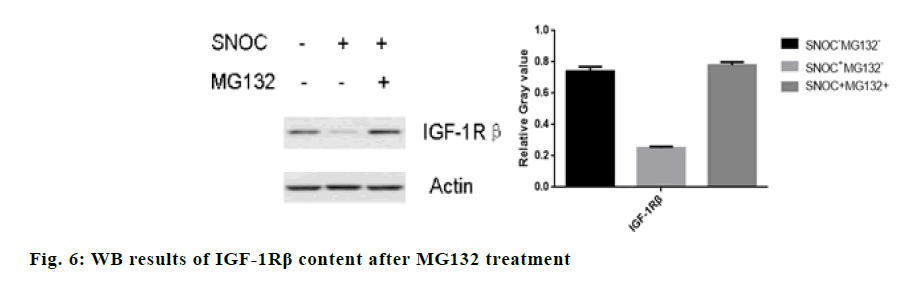
The IGF-1Rβ level in hippocampal neuron cells after treatment with proteasome inhibitor MG132 was significantly higher than cells free from MG132 (p<0.05). fig. 6 shows that MG132 can reverse the degradation of IGF-1Rβ induced by SNOC, indicating that proteasome pathway participates in the degradation of IGF-1Rβ induced by SNOC.
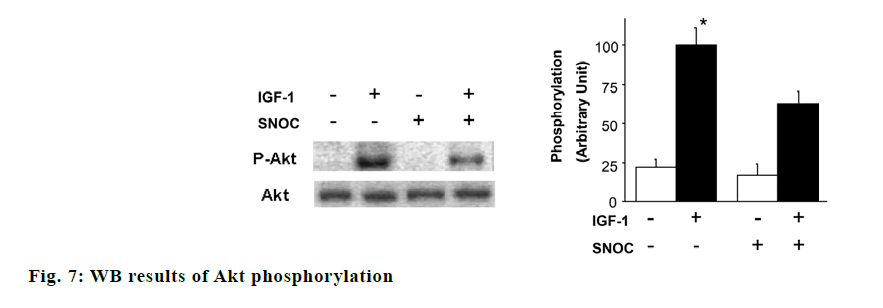
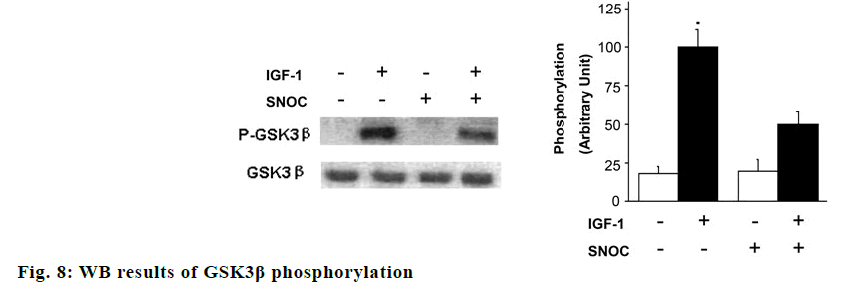
The effects of SNOC and IGF-1 on the phosphorylation of AKT, GSK3β and tau protein were showed in fig. 7. Akt phosphorylation and GSK3β phosphorylation in hippocampal neuron cells treated with SNOC decreased significantly compared to that of cells treated without SNOC (p<0.05), while Tau phosphorylation level in hippocampal neuron cells treated with IGF-1 increased significantly compared with that of cells free from IGF-1 (p<0.05). As shown in fig. 8, the phosphorylation degree of GSK3β in neurons cells treated with SNOC decreased significantly (p<0.05), suggested that NO pathway may be involved in the signal transmission of IGF-1 pathway.
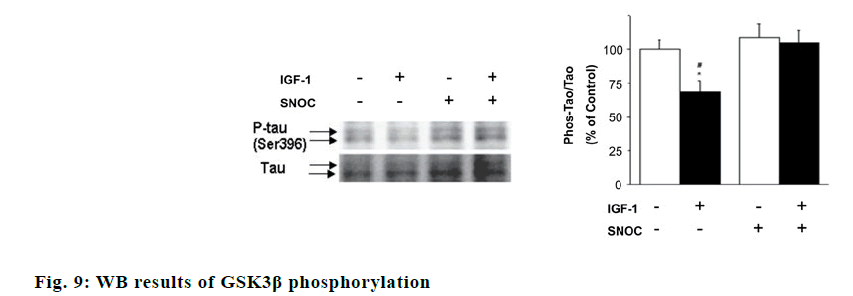
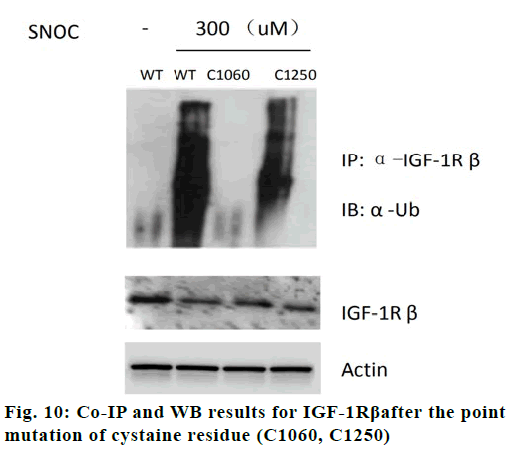
As shown fig. 9, IGF-1 can reduce tau protein phosphorylation in neurons (p<0.05). When SNOC is given to incubate neurons in advance, it can improve Tau protein phosphorylation (p<0.05) in neurons cells, which indicates that NO pathway may participate in the effect of IGF-1 pathway on tau protein phosphorylation. As shown in fig. 10, SNOC increased the ubiquitination of IGF-1Rβ and decreased the expression of IGF-1Rβ (p<0.05). The C1060 mutation in the intracellular domain of IGF-1Rβ reduced the ubiquitination of IGF-1Rβ by SNOC and increased the IGF-1Rβ expression, which shows that C1060 site is necessary for its ubiquitination and degradation.
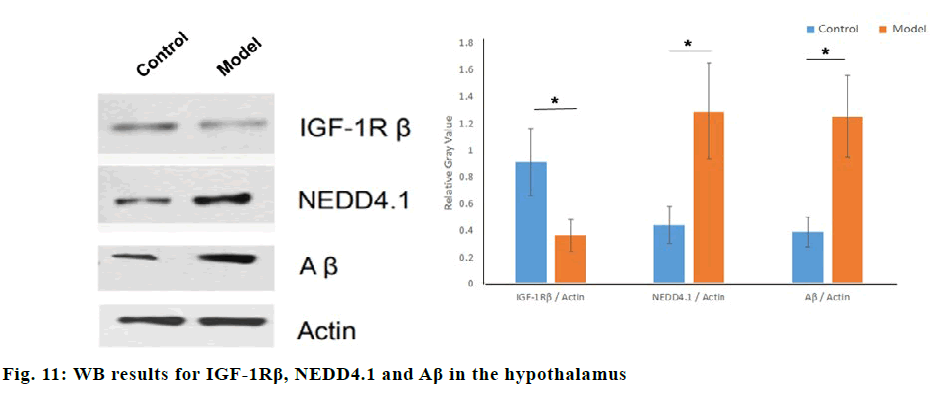
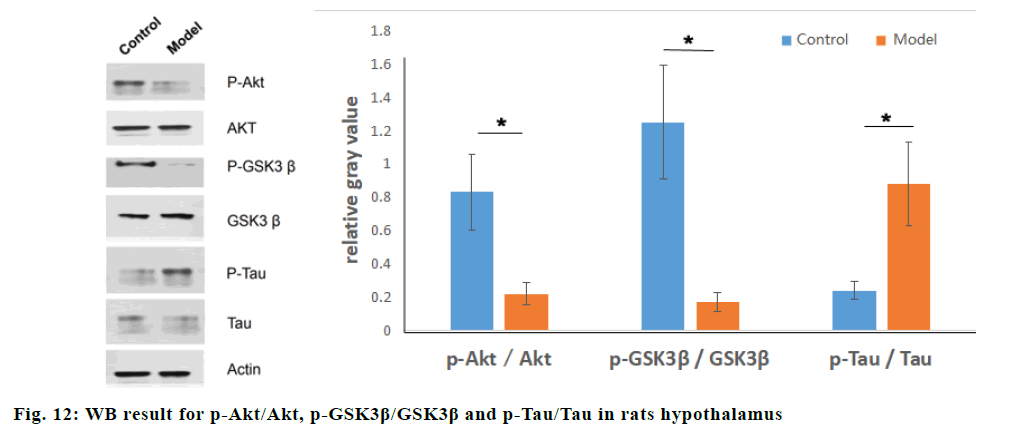
Western blot was used to compare the relative gray values of IGF-1Rβ, NEDD4.1, β-amyloid (Aβ) in normal rats and STZ dementia models. Compared with the normal group, the relative gray values of IGF-1Rβ in the model group were significantly lower (p<0.05), and the relative gray values of NEDD4.1 and Aβ were significantly higher (p<0.05), as shown in fig. 11. IGF-1Rβ expression was negatively correlated with NEDD4.1 by Pearson correlation analysis(r=-0.92, p=0.03). Compared with the normal group, the relative gray values of p-Akt/Akt and p-GSK3β/GSK3β in the STZ dementia group were significantly lower (p<0.05), and the relative gray values of p-Tau/Tau were significantly increased (p<0.05), as shown in fig. 12.
AD has become a major challenge for aging society worldwide. The typical pathological changes include senile plaques, neurofibrillary tangles and neuron loss.
Some scholars called AD as “type 3 diabetes” or “brain type diabetes”, for AD patients generally occur abnormal insulin signaling pathway in the brain, which is called “insulin resistant brain state” [10]. The characteristics of insulin resistance in peripheral tissues have been widely recognized, but the abnormality of central insulin signalling pathway and its mechanism are controversial, especially its role in the pathological process of AD. Compared with the decrease of glucose utilization in AD patients, advanced glycation products induced by hyperglycemia accumulated in the senile plaques and nerve fibre tangles in AD brain. Insulin receptor (IR) and IGF-1R are two kinds of essential receptor tyrosine kinases. After binding with corresponding ligands, they induce self-phosphorylation, which promote a series of intracellular molecules including insulin receptor substrate (IRS-1 and IRS-2) phosphorylation, and then activate different signalling pathways [11]. Insulin signaling system is involved in the regulation of Aβ synthesis through PI3K/Akt, MAPK, Wnt and other signalling pathways, promotes the extracellular transport of Aβ and regulates Tau protein phosphorylation, and is closely related to neuron apoptosis and regeneration, learning and memory, and cognitive function [12], impaired insulin signal system leads to the increase of glycogen synthesis kinase-3β activity and the abnormal phosphorylation of Tau protein. When insulin level in the brain increases, it can also compete with Aβ for insulin degrading enzyme (IDE), which can degrade insulin and Aβ at the same time, resulting in Aβ degradation and aggregation. Therefore, the abnormality of insulin signalling pathway has an important effect on two key proteins Aβ and Tau in the pathological characteristics of AD, and its role in the pathological process of AD has become one of the research hotspots nowadays.
In this study, we isolated and cultured the hippocampal neurons cells from ICH mice firstly. As SNOC can release a large dose of NO, we used SNOC to induce the neurotoxic injury to hippocampal neurons cells as in vitro model. The Western Blot results showed that SNOC could increase the ubiquitination of IGF-1Rβ in a dose-dependent manner and decrease the expression of IGF-1Rβ in vitro brain injury cells, which proved that the in vitro brain injury model that can increase ubiquitination of IGF-1Rβ was successfully constructed. Moreover, according to our study, NEDD4.1 increased in injury model, indicating that NEDD4.1 was positively correlated with the ubiquitination of IGF-1Rβ in brain injured cells.
In order to further confirm the effect of NEDD4.1 on the ubiquitination of IGF-1Rβ in AD patients, NEDD4.1 was up-regulated by pCMV-NEDD4.1 vector or down regulated by siRNA in mouse hippocampal neurons ed following SNOC treatment. The results showed that after over-expression of NEDD4.1, the ubiquitination level of IGF-1Rβ increased significantly. After silencing NEDD4.1 gene, the ubiquitination of IGF-1Rβ decreased, and the inhibition of IGF-1Rβ expression by SNOC was reversed. These results suggest that the ubiquitination of IGF-1Rβ by NEDD4.1 may be the key mechanism of IGF-1Rβ degradation.
In order to study the mechanism of NEDD4.1 on the ubiquitination of IGF-1Rβ, cyclohexide was added to inhibit the synthesis of new proteins. It was found that SNOC can still induce the degradation of IGF-1Rβ in a time-dependent manner, which indicates that the IGF- 1Rβ level in damaged brain tissues is less relative to protein synthesis. MG132 can reverse the degradation of IGF-1Rβ induced by SNOC, indicating that the proteasome pathway is involved in the degradation of IGF-1Rβ induced by SNOC.
Then, we studied the effect of SNOC treatment in vitro on IGF-1-induced phosphorylation of Akt and GSK3β in cultured neurons, which are crucial molecules of IGF-1 pathway [13]. Phosphorylation of Akt and GSK3β after 10 nM IGF-1 stimulation for 5 min are indicated. Pretreatment with 200 μM SNOC can decrease the phosphorylation of Akt and GSK3β induced by IGF-1. The mutation of C1060 site in intracellular domain of IGF-1Rβ reduced the ubiquitination of IGF- 1Rβ by SNOC and increased the expression of IGF- 1Rβ, which indicated that C1060 site was necessary for its ubiquitination and degradation.
In order to study the mechanism of NEDD4.1 on the ubiquitination of IGF-1Rβ in vivo, STZ injected into the lateral ventricles of rats to interfer with the insulin signal pathway in neurons, which would lead to the decrease of the ability of spatial learning and memory in rats. A dementia model of insulin resistance was constructed. The results of animal experiments showed that STZ reduced IGF-1Rβ level while enhanced secretion of NEDD4.1 and Aβ in brain. Meanwhile, STZ inhibited the activity of IGF-1 signal pathway, decreasing the phosphorylation of Akt and GSK3β, and increased the phosphorylation level of Tau protein. The results suggested that NEDD4.1 may influence Tau and Aβ by IGF-1 signal pathway on account of rising of phosphorylation of Akt and GSK3β.
In conclusion, our in vivo and in vitro experiments showed that abnormal increasing NEDD4.1 in brain injury tissue induced the rising ubiquitination of IGF-1Rβ. C1060 site in IGF-1Rβ was necessary for the NEDD4.1 of ubiquitination and degradation of IGF-1Rβ. In addition, NEDD4.1 affects IGF-1 signal pathway in brain, such as inhibiting Akt and GSK3β phosphorylation, promoting Tau protein phosphorylation, and promoting Aβ secretion.
Authors’ contributions
MIN YE conceived and designed the experiments; XINGJUN WU, LIHUA CHEN, LIWEN ZHANG, YINGFANG ZHANG and CHENYANG ZHANG performed the experiments; HUAN GAO analyzed the data and wrote the paper.
Acknowledgement
This work was supported by Affiliated BenQ Hospital of Nanjing Medical University.
Conflicts of interest
The authors report no conflicts of interest.
References
- Rallabandi VS, Tulpule K, Gattu M. Alzheimer's Disease Neuroimaging Initiative. Automatic classification of cognitively normal, mild cognitive impairment and Alzheimer's disease using structural MRI analysis. Informatics in Medicine Unlocked 2020;27:100305.
- Niewiadomska G, Mietelska-Porowska A, Mazurkiewicz M. The cholinergic system, nerve growth factor and the cytoskeleton. Behav Brain Res 2011;221:515-26.
- Brown BM, Peiffer J, Rainey-Smith SR. Exploring the relationship between physical activity, beta-amyloid and tau: A narrative review. Ageing Res Rev 2019;50:9-18.
- Singulani MP, Pereira CP, Ferreira AF, Garcia PC, Ferrari GD, Alberici LC, et al. Impairment of PGC-1α-mediated mitochondrial biogenesis precedes mitochondrial dysfunction and Alzheimer's pathology in the 3xTg mouse model of Alzheimer's disease. Exp Gerontol 2020;133:110882.
- Allnutt MA, Jacobson S. Do herpesviruses play a role in Alzheimer's disease pathogenesis? Drug Discov Today Dis Models 2019;9:76-84.
- Akhtar A, Sah SP. Insulin signaling pathway and related molecules: Role in neurodegeneration and Alzheimer's disease. Neurochem Int 2020;135:104-7.
- De Felice FG, Lourenco MV, Ferreira ST. How does brain insulin resistance develop in Alzheimer's disease?. Alzheimers Dement 2014;10:26-32.
- Moloney AM, Griffin RJ, Timmons S, O’Connor R, Ravid R, O’Neill C. Defects in IGF-1 receptor, insulin receptor and IRS-1/2 in Alzheimer's disease indicate possible resistance to IGF-1 and insulin signalling. Neurobiol Aging 2010;31:224-43.
- Semelakova M, Grauzam S, Betadthunga P, Tiedeken J, Coaxum S, Neskey DM, et al. Vimentin and Non-Muscle Myosin IIA are Members of the Neural Precursor Cell Expressed Developmentally Down-Regulated 9 (NEDD9) Interactome in Head and Neck Squamous Cell Carcinoma Cells. Transl Oncol 2019;12:49-61.
- Kandimalla R, Thirumala V, Reddy PH. Is Alzheimer's disease a Type 3 Diabetes? A critical appraisal. Biochim Biophys Acta 2017;1863:1078-89.
- Johansson P, Åberg D, Johansson JO, Mattsson N, Hansson O, Ahrén B, et al. Serum but not cerebrospinal fluid levels of insulin-like growth factor-I (IGF-I) and IGF-binding protein-3 (IGFBP-3) are increased in Alzheimer's disease. Psychoneuroendocrinology 2013;38:1729-37.
- Manolopoulos KN, Klotz LO, Korsten P, Bornstein SR, Barthel A. Linking Alzheimer's disease to insulin resistance: the FOXO response to oxidative stress. Mol psychology 2010;15:1046-52.
- Mohamed WA, Salama RM, Schaalan MF. A pilot study on the effect of lactoferrin on Alzheimer`s disease pathological sequelae: Impact of the p-Akt/PTEN pathway. Biomed Pharmacother 2019;111:714-23.