- *Corresponding Author:
- X. Liu
Department of Otorhinolaryngology,
Hunan Provincial People’s Hospital,
Changsha,
Hunan 410005,
China
E-mail: 736457144@qq.com
| This article was originally published in a special issue, “Trending Topics in Biomedical Research and Pharmaceutical Sciences” |
| Indian J Pharm Sci 2022:84(1) Spl Issue “284-289” |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
Laryngeal cancer has become the focus of research because of its high incidence rate and mortality rate. However, the research on microRNA-194-5p in laryngeal cancer is quite rare. The purpose of this study is to explore the effect of microRNA-194-5p on the proliferation and invasion of laryngeal cancer cells, in order to find an effective way to treat laryngeal cancer. The results showed that microRNA-194-5p could activate mammalian target of rapamycin signaling pathway by regulating the expression of smad ubiquitin regulatory factor 1, which had an effect on laryngeal cancer cells. The results showed that the absorbance of microRNA-194-5p group was 0.38 lower than that of negative control group, which indicated that up regulation of microRNA-194-5p could weaken the proliferation of laryngeal cancer cells. In addition, the average number of laryngeal cancer cells in negative control group and microRNA-194-5p group was 125.2 and 53.8 respectively, which indicated that microRNA-194-5p could reduce the number of laryngeal cancer cells passing through basement membrane and their invasion ability.
Keywords
Radiotherapy, laryngeal cancer, microRNA-194-5p, cell proliferation, Western blotting
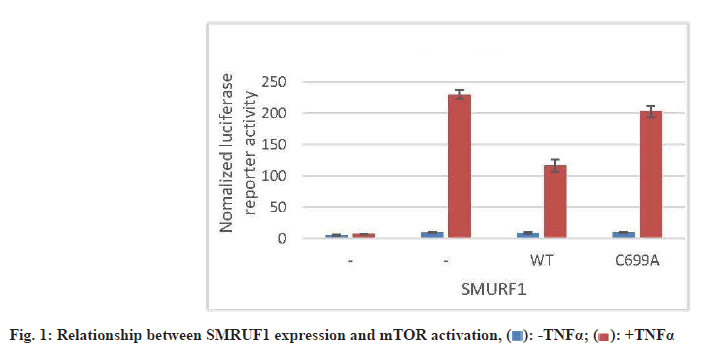
Laryngeal carcinoma main pathological type is squamous cell carcinoma. At present, the treatment of laryngeal cancer is mainly surgery, radiotherapy and chemotherapy. However, there are many complications and adverse reactions in surgery and radiotherapy and chemotherapy. Recurrence and metastasis after treatment will still lead to death of laryngeal cancer patients. It is very important to find a new and effective treatment for laryngeal cancer. Therefore, the treatment of laryngeal cancer by interfering with the proliferation and invasion of cancer cells has become the focus of this study. The research on the treatment of laryngeal cancer is the focus of the medical staff and researchers all over the world, and the research results are endless. For example, Xuan studied the expression of circular RNA (circRNA) in four pairs of matched lung squamous cell carcinoma tissues and adjacent non tumor tissues by microarray analysis[1]. Further detection of HSA by quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)-circRNA-100855 is the most upregulated circRNA, HSA-circRNA-104912 is the downregulated circRNA[2-5]. Peretti studied the reasonable limit of transoral laser microsurgery in the treatment of laryngeal cancer[6-8]. To highlight the most appropriate and repeatable indications and to integrate this treatment tool into the overall framework of alternative treatment strategies, such as open partial laryngectomy and nonsurgical organ preservation protocol[9]. Manuel introduced in detail the infiltration of laryngeal cartilage tumor through the whole series of organ sections, in which the infiltration was regarded as horizontal infiltration (through cartilage) and vertical infiltration (degree of infiltration)[10-12]. After understanding the target gene and regulatory mechanism of miR-194-5p, scholars all over the world are actively studying the corresponding therapeutic effect[13,14]. Apoptosis can be detected by fluorescence activated cell sorting and insulin-like growth factor-1 receptor protein level of miR-194-5p target gene can be detected by Western blotting[15-17]. Hu, the miR analysis of 48 breast cancer patients’ sera was carried out by exiqon mercury miR RT-PCR[18]. Seven miRNAs differentially expressed in recurrent and non-recurrent breast cancer patients were identified[19,20]. MiR-194-5p and miR-122 were down regulated in human glioma and glioma stem cells. MiR- 194-5p[21,22]. Their research provides a reference for this study and avoids the mistakes in their research[23]. The proliferation ability, cycle and invasion ability of laryngeal cancer cells were analyzed by transfection, protein extraction test and cell proliferation test. The results showed that 97 % of the experimental samples showed that miR-194-5p could affect the proliferation and invasion of laryngeal cancer cells by regulating the expression of Smad ubiquitin regulatory factor 1 (SMURF1) and activating the mammalian Target of Rapamycin (mTOR) signal pathway. The laryngeal cancer tissue and adjacent tissue of 36 patients with laryngeal cancer in a cancer hospital were collected and stored in liquid nitrogen. Mir-194-5p agomir and Negative Control (NC) dilution reagent; cell culture reagent, including 0.01 M Phosphate Buffered Saline (PBS) (pH 7.4) and 90 % serum cell cryopreservation solution; Western blot reagent. The experimental instruments include biosafety cabinet, cell incubator, ultra-low temperature refrigerator, SpectraMax 190 full wavelength reader, automatic autoclave, micro pipette gun, pure water filter, desktop centrifuge, etc. First, 10 ml complete culture medium including 16 % fetal bovine serum, 100 U/ml penicillin and 100 U/ml deoxymycin were added into the culture bottle. Then, the cryopreservation tube was quickly transferred from liquid nitrogen to a water bath at 37°. The cryopreservation was continuously moved in the water bath and the cell suspension was thawed within 1 min as much as possible. Wipe the cryopreservation tube with alcohol gauze for sterilization and transfer the cell suspension in the cryopreservation tube to the culture bottle containing the culture medium. After the cell adheres to the wall, the cells are treated with liquid exchange to remove the toxic effect of Dimethyl Sulfoxide (DMSO) on the cells. The resuscitated laryngeal cancer cells were cultured regularly in the medium at saturated humidity. In the process of cell culture, we should pay attention to the color change of the culture medium, observe the change of cell morphology and cell fusion degree with the inverted microscope and replace the new culture medium in time to keep the cell in a good growth state. When the degree of cell fusion reaches about 80 %, cells are propagated. The human larynx epithelioma cancer (HEp-2) cells in logarithmic growth period were inoculated into 24 well culture plate with 1000 cells/ml, 1 ml per well. Under the normal condition, change the solution every 3 d for 10 d. To observe the cell death, select the lowest concentration that can kill cells completely on the 10th d as the screening concentration of the transfected cell resistance gene. The concentration of G418 was 800 μg/ml. Lipofectamine 2000 was mixed with the culture medium and then added to the 24 well culture plate. The experiment was continued 24 h after transfection. The experiment was divided into three groups: Blank control group, miR-194-5p group and miR-194-5p NC group. The cells of each group were collected by digestion and centrifugation, washed for three times with PBS solution, added 100 μl of cell lysate, split on ice, shake and mix every 10 min, split for 30 min, collected the cells in 1.5 ml Eppendorf® (EP) tube, centrifuged for 15 min at 4°, absorbed the supernatant of protein and stored separately. The protein concentration was determined according to the instructions of Bicinchoninic Acid (BCA) white concentration test kit. First, the standard protein was prepared into a concentration of 0.5 mg/ml and then the corresponding amount of BCA working solution was prepared according to the number of samples to be tested (the A solution and B solution in the kit were mixed in the proportion of 50:1). Add 100 μl of test solution into each hole of 96 hole plate and mix the diluted standard and the sample to be tested. Incubate in 37° incubator for 30 min, cool down and measure the absorbance with enzyme standard instrument. Draw the standard curve of protein concentration and calculate the actual protein concentration of each sample. HEp-2, HEp-2/mock and HEp-2/Tiaml cells in the logarithmic growth stage were digested and resuspended by trypsin and then 1000 cells were inoculated into 96 well plates, with a volume of 1 ml per well. Each group of cells had four subsidiary pores and a blank control (only medium was added). Then 3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyl-2H-tetrazolium bromide (MTT) method was used to detect the growth of cells in each group every 24 h for 3 consecutive days. At the time point, 20 μl of 3 mg/ml MTT solution was added into each pore and the culture was continued in 37° cell incubator for 4 h and then the culture was terminated. After carefully sucking and discarding the medium in the hole with a 1 ml syringe, add 100 μl DMSO into each hole. Shake the l min on the Wellscan Mk 2 washing machine to make the crystal fully dissolved, adjust the blank control pore to zero and measure the absorbance value (Optical Density (OD) value) of each pore at 570 nm of the enzyme-linked immunosorbent detector. The experiment was repeated three times. The average value of the results was taken and the corresponding OD value was used to represent the cell proliferation capacity and the growth curve was drawn[24,25]. After 24 h of transfection, the laryngeal cancer cells of miR-194- 5p group and NC group were collected in 1.5 ml EP tube. The cells were pretreated with starvation and the number of cells could not be less than 1×106. 70 % ethanol was fixed at 4° overnight, then 100 μl RNase A was added and incubated at 37° for 30 min, then Propidium Iodide (PI) staining was added at 4° for 30 min (dark) and then on-board detection was carried out[26]. Since mTOR was identified, its related research has been a hot research direction in the field of life science in recent years[27,28]. Because mTOR signaling pathway is involved in the regulation of many physiological functions in the body, its disorder will directly lead to inflammation and even cancer, so it’s fine regulation is very important for the maintenance of homeostasis and the in-depth excavation of its regulation mechanism is also very important[29]. Therefore, we want to know whether SMURF1, a member of the heat family, can regulate the mTOR signaling pathway. First, we tested whether the overexpressed SMURF1 would affect the activation of mTOR. The E3 ubiquitin ligase SMURF1 (Wild Type (WT) and c699a) was overexpressed in laryngeal cancer cells. The effect of SMURF1 on mTOR activation was observed after Tumor Necrosis Factor alpha (TNF-α) induced activation. As shown in fig. 1, only SMURF1- WT activated TNF-α to induced mTOR, while SMURF1-c699a had no significant effect. Moreover, our experiment further confirmed that the activation effect will be more significant with the increase of SMURF1 expression. Cancer cells can proliferate infinitely. They ignore the signals of normal proliferation control and only follow their own internal proliferation standards. They can even move and invade adjacent organizations. MiR-194-5p can affect the activation of mTOR signaling pathway by regulating the expression of SMURF1, thus affecting the proliferation of laryngeal cancer cells. Rapid quantification of miR discovery is the key to the elimination of miR in gene regulation. At present, the detection methods of miR mainly include northern blotting, microarray analysis gene chip technology (microarray), real-time fluorescence quantitative PCR. High throughput analysis cannot be performed due to the low sensitivity of northern blotting. In general, qRT-PCR is used to confirm the predicted relative performance level of miRNA. Gene chip has the characteristics of high throughput, high precision and fast detection. It is widely used in gene diagnosis and experimental research[30]. The stronger the proliferation of laryngeal cancer cells, the more difficult it is to inhibit and the higher the harm to human body. As shown in fig. 2, compared with NC group and blank control group, the absorbance of miR-194-5p group cells decreased significantly at 24 h, 48 h and 72 h. Cancer cells also have cell cycle, different cytotoxic drugs act on different stages of cancer cell division to prevent its division, so as to play an anti-tumor role. There are two mechanisms of cell cycle regulation, including cell cycle driving mechanism and regulatory mechanism. When the regulatory mechanism of cell cycle is destroyed and transform into tumor cells. The cell cycle can be divided into four successive periods. That is G1, S, G2 and M. G1 phase refers to the gap period from the completion of the last mitosis to the DNA replication that is the preparation period for cell growth and DNA synthesis. The length of this phase varies with cells. In the cell cycle, the genetic information is precisely duplicated by DNA replication of genome and equal separation of genetic information of M phase and transferred from mother cell to two daughter cells. G2 stage and mitosis entry: G2 stage refers to the time interval from the completion of DNA replication to the beginning of mitosis. Mitosis: Usually divided into prophase, metaphase, anaphase, telophase and cytokinesis. Flow cytometry was used to detect and record the cell cycle of laryngeal cancer in NC group and miR-194-5p group. The results are shown in Table 1. The percentage of G0/G1 cells in miR-194-5p group was higher than that in NC group (p<0.01). This suggests that up regulation of miR-194-5p can slow down the progression of laryngeal cancer cell cycle. The invasion in the late stage of tumor, which is the result of high deterioration of tumor lesions and high development of malignant behavior. It is a sign of advanced malignant tumor. Cancer cells are unicellular like organisms, whose invasion includes invasive tumor cells invading other adjacent tissues, degeneration and necrosis of the invading tissues and progressive changes in space and time. The reason for the invasion is: First, because cancer cells have the ability of amoeba movement, they can extend pseudopodia to insert into the surrounding tissue gap, run and destroy the surrounding tissue or even replace it. Another reason is that many cancer cells can also secrete some special molecular substances to help cancer cells adhere to the surrounding tissues. If the number of cells is small, it indicates that the invasion ability is weak; otherwise, it indicates that the invasion ability is strong. As shown in fig. 3, the number of cells passing through the basement membrane in miR-194-5p group was significantly lower than that in NC group, which indicated that up regulating the expression of miR-194-5p could inhibit the over expression of SMURF1, activate the mTOR signal pathway and reduce the invasion ability of laryngeal cancer cells. Laryngeal cancer is one of the common tumor diseases, early diagnosis and treatment can improve the survival rate of patients, but for late patients, laryngeal cancer often occurs metastasis, the metastasis of cancer cells is mainly related to the proliferation and invasion of cancer cells. In this paper, the relationship between miR-194-5p and laryngeal cancer was studied and discussed, and the proliferation, cycle and invasion ability of mir-194-5p and pachytene cells were compared. MiR-194-5p can not only be used as a new diagnostic marker of laryngeal cancer, to detect the possibility of metastasis of laryngeal cancer, but also be used to open up a new way of treatment of laryngeal cancer. Through miR-194-5p, the proliferation of laryngeal cancer cells can be interfered, the invasion ability can be reduced and the disease of patients with laryngeal cancer can be alleviated. This brings a new dawn for the treatment and research of laryngeal cancer and other tumor diseases.
| Group | n | Proportion of cells in each stage (%) | ||
|---|---|---|---|---|
| G0/G1 | S | G2/M | ||
| NC Group | 5 | 50.88±2.11 | 41.71±1.23 | 7.41±0.2 |
| miR-194-5p Group | 5 | 62.74±2.36 | 30.35±1.98 | 8.91±0.19 |
Note: G0: Resting state; G1: Growth 1 phase; G2: Growth 2 phase; S: Synthesis phase; M: Mitotic phase
Conflict of interests:
The authors declared no conflict of interest.
References
- Xuan L, Qu L, Zhou H, Wang P, Yu H, Wu T, et al. Circular RNA: A novel biomarker for progressive laryngeal cancer. Am J Transl Res 2016;8(2):932-9.
- Smith MM, Abrol A, Gardner GM. Assessing delays in laryngeal cancer treatment. Laryngoscope 2016;126(7):1612-5.
[CrossRef] [Google Scholar] [Pub Med]
- Shivappa N, Hébert JR, Rosato V, Serraino D, La Vecchia C. Inflammatory potential of diet and risk of laryngeal cancer in a case–control study from Italy. Cancer Causes Control 2016;27(8):1027-34.
[CrossRef] [Google Scholar] [Pub Med]
- Luo X, Tang M, Hu Q. Genomic DNA methylation of non-small cell lung cancer applied in early screening of tumor. Indian J Pharm Sci 2020:31-5.
- Klobučar M, Sedić M, Gehrig P, Grossmann J, Bilić M, Kovač-Bilić L, et al. Basement membrane protein ladinin-1 and the MIF-CD44-β1 integrin signaling axis are implicated in laryngeal cancer metastasis. Biochim Biophys Acta 2016;1862(10):1938-54.
[CrossRef] [Google Scholar] [Pub Med]
- Peretti G, Piazza C, Mora F, Garofolo S, Guastini L. Reasonable limits for transoral laser microsurgery in laryngeal cancer. Curr Opin Otolaryngol Head Neck Surg 2016;24(2):135-9.
[CrossRef] [Google Scholar] [Pub Med]
- Zeng YC, Xing R, Zeng J, Xue M, Chi F, Xin Y, et al. Sodium glycididazole enhances the radiosensitivity of laryngeal cancer cells through downregulation of ATM signaling pathway. Tumor Biol 2016;37(5):5869-78.
[CrossRef] [Google Scholar] [Pub Med]
- Zuo J, Wen J, Lei M, Wen M, Li S, Lv X, et al. Hypoxia promotes the invasion and metastasis of laryngeal cancer cells via EMT. Med Oncol 2016;33(2):1-9.
[CrossRef] [Google Scholar] [Pub Med]
- Regeling B, Laffers W, Gerstner AO, Westermann S, Müller NA, Schmidt K, et al. Development of an image pre‐processor for operational hyperspectral laryngeal cancer detection. J Biophotonics 2016;9(3):235-45.
[CrossRef] [Google Scholar] [Pub Med]
- Gómez Serrano M, Iglesias Moreno MC, Gimeno Hernández J, Ortega Medina L, Martín Villares C, Poch Broto J. Cartilage invasion patterns in laryngeal cancer. Eur Arch Otorhinolaryngol 2016;273(7):1863-9.
[CrossRef] [Google Scholar] [Pub Med]
- Peng WJ, Mi J, Jiang YH. Asbestos exposure and laryngeal cancer mortality. Laryngoscope 2016;126(5):1169-74.
[CrossRef] [Google Scholar] [Pub Med]
- Yu H, Lu Q, Wang Y, Ma X, Li B, Yang L. Differential Expression of ANXA2 in Eutopic and Ectopic Endometrium. Indian J Pharm Sci 2020:35-9.
- Zhang Z, Lei B, Wu H, Zhang X, Zheng N. Tumor suppressive role of miR-194-5p in glioblastoma multiforme. Mol Med Rep 2017;16(6):9317-22.
[CrossRef] [Google Scholar] [Pub Med]
- Jiang MJ, Chen YY, Dai JJ, Gu DN, Mei Z, Liu FR, et al. Dying tumor cell-derived exosomal miR-194-5p potentiates survival and repopulation of tumor repopulating cells upon radiotherapy in pancreatic cancer. Mol Cancer 2020;19(1):1-5.
- Samandari N, Mirza AH, Nielsen LB, Kaur S, Hougaard P, Fredheim S, et al. Circulating microRNA levels predict residual beta cell function and glycaemic control in children with type 1 diabetes mellitus. Diabetologia 2017;60(2):354-63.
[CrossRef] [Google Scholar] [Pub Med]
- Su R, Cao S, Ma J, Liu Y, Liu X, Zheng J, et al. Knockdown of SOX2OT inhibits the malignant biological behaviors of glioblastoma stem cells via up-regulating the expression of miR-194-5p and miR-122. Mol Cancer 2017;16(1):1-22.
[CrossRef] [Google Scholar] [Pub Med]
- Jiang Y, Gao H, Liu M, Mao Q. Sorting and biological characteristics analysis for side population cells in human primary hepatocellular carcinoma. Am J Cancer Res 2016;6(9):1890-1905.
- Huo D, Clayton WM, Yoshimatsu TF, Chen J, Olopade OI. Identification of a circulating microRNA signature to distinguish recurrence in breast cancer patients. Oncotarget 2016;7(34):55231-48.
[CrossRef] [Google Scholar] [Pub Med]
- Jung DE, Park SB, Kim K, Kim C, Song SY. CG200745, an HDAC inhibitor, induces anti-tumour effects in cholangiocarcinoma cell lines via miRNAs targeting the Hippo pathway. Sci Rep 2017;7(1):1-3.
- Farré PL, Scalise GD, Duca RB, Dalton GN, Massillo C, Porretti J, et al. CTBP1 and metabolic syndrome induce an mRNA and miRNA expression profile critical for breast cancer progression and metastasis. Oncotarget 2018;9(17):13848-58.
[CrossRef] [Google Scholar] [Pub Med]
- Lu Y, Zhang Y, Lu Q, Li Z, Xie X. Differential expression profiles assay of miRNAs in rat serum after traumatic hemorrhagic shock. Int J Clin Exp Pathol 2016;9(12):12262-74.
- Ninomiya M, Kondo Y, Kimura O, Funayama R, Nagashima T, Kogure T, et al. The expression of miR‐125b‐5p is increased in the serum of patients with chronic hepatitis B infection and inhibits the detection of hepatitis B virus surface antigen. J Viral Hepat 2016;23(5):330-9.
[CrossRef] [Google Scholar] [Pub Med]
- Liu X, He Y, Cao X, Liu L, Tan H. Effect of dezocine combined with dexmedetomidine on postoperative pain and quality of life in patients with advanced liver cancer. Indian J Pharm Sci 2020:36-40.
- Lee YS, Park Y, Kwon M, Roh JL, Choi SH, Nam SY, et al. Expression of lysyl oxidase predictive of distant metastasis of laryngeal cancer. Otolaryngol Head Neck Surg 2017;156(3):489-97.
[CrossRef] [Google Scholar] [Pub Med]
- Tuomi L, Johansson M, Andréll P, Finizia C. Interpretation of the swedish self-evaluation of communication experiences after laryngeal cancer: Cutoff levels and minimum clinically important differences. Head Neck 2016;38(5):689-95.
[CrossRef] [Google Scholar] [Pub Med]
- Zhu D, Wang L. Effects of carbachol on the excitatory muscarinic cholinergic receptors in urinary bladder and plasma inflammatory mediators in anesthetized rats. Indian J Pharm Sci 2020;82(1):38-42.
- Sun C, Han X, Li X, Zhang Y, Du X. Diagnostic performance of narrow band imaging for laryngeal cancer: A systematic review and meta-analysis. Otolaryngol Head Neck Surg 2017;156(4):589-97.
[CrossRef] [Google Scholar] [Pub Med]
- Lu E, Su J, Zhou Y, Zhang C, Wang Y. CCL20/CCR6 promotes cell proliferation and metastasis in laryngeal cancer by activating p38 pathway. Biomed Pharmacother 2017;85:486-92.
[CrossRef] [Google Scholar] [Pub Med]
- Klatka J, Grywalska E, Hymos A, Guz M, Polberg K, Roliński J, et al. Cyclooxygenase-2 inhibition enhances proliferation of NKT cells derived from patients with laryngeal cancer. Anticancer Res 2017;37(8):4059-66.
[CrossRef] [Google Scholar] [Pub Med]
- Wang N, Liu X. The application of enoxaparin sodium injection in the treatment of cerebral infarction. Indian J Pharm Sci 2020;82(2):39-44.
 ): -TNFα; (
): -TNFα; (  ): +TNFα
): +TNFα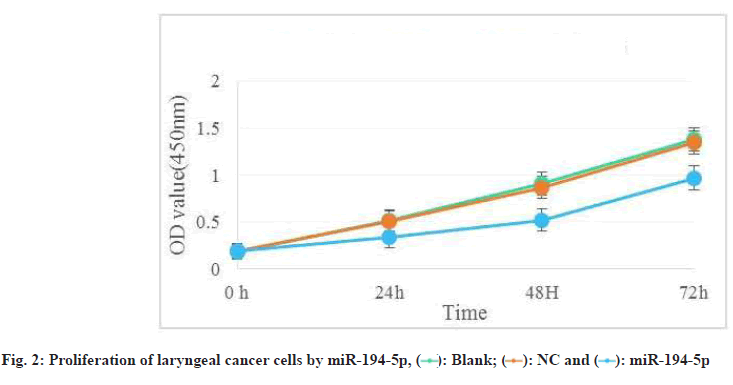
 ): Blank; (
): Blank; (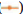 ): NC and (
): NC and ( ): miR-194-5p
): miR-194-5p 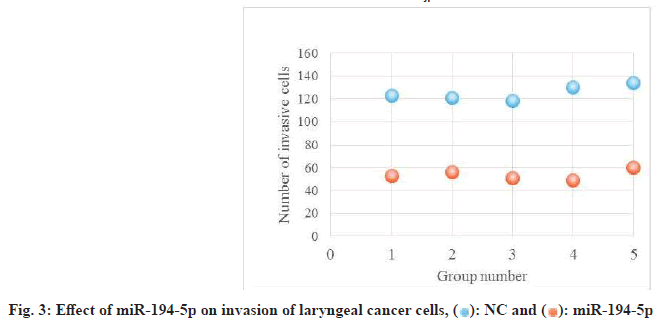
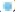 ): NC and (
): NC and ( ): miR-194-5p
): miR-194-5p 



